Lactobacillus Species from Iranian Jug Cheese: Identification and Selection of Probiotic Based on Safety and Functional Properties
Applied Food Biotechnology,
Vol. 8 No. 1 (2021),
14 December 2020
,
Page 47-56
https://doi.org/10.22037/afb.v8i1.29253
Abstract
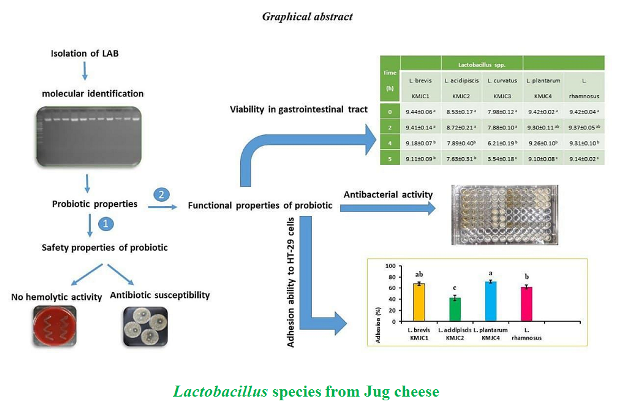
Background and Objective: Traditional fermented products are appropriate sources for the isolation of indigenous bacteria with probiotic characteristics and potential similar or better than commercial probiotics. In this study, Lactobacillus species were isolated from jug cheese, a type of Iranian traditional cheese, and their potential probiotic characteristics were studied.
Material and Methods: Study of the probiotic species included hemolytic activity, antibiotic susceptibility, inhibitory activity against pathogenic bacteria, low pH and bile salts tolerance, viability in gastrointestinal tract conditions and adhesion ability to HT-29 cells.
Results and Conclusion: Results showed that the isolates included no hemolytic activity and were susceptible or intermediate susceptibility to most antibiotics. Of four isolates, Lacto-bacillus plantarum KMJC4 showed the strongest antibacterial activity (MIC = 6.25 mg ml-1) against Escherichia coli, Staphylococcus aureus, Listeria monocytogenes, Bacillus cereus and Salmonella enterica subsp. enterica serovar Typhimurium. All the isolates, except Lactobacillus curvatus KMJC3, preserved their viability after transition through the simulated gastrointestinal tract conditions above 106 CFU ml-1. Lactobacillus acidipiscis KMJC2 and Lactobacillus plantarum KMJC4 showed the lowest and the highest adhesion rates to HT-29 cells with 3.55 and 6.80 Log10 CFU ml-1 (42.51 and 71.35%), respectively. Lactobacillus plantarum KMJC4 included a better bacterial inhibitory activity and adhesion to HT-29 cells than that Lactobacillus rhamnosus GG did as control. Lactobacillus brevis KMJC1 demonstrated appropriate probiotic characteristics such as antibacterial activity, viability in low pH, bile salts and gastrointestinal tract conditions and adhesion capability to HT-29 cells. In conclusion, Lactobacillus plantarum KMJC4 and Lactobacillus brevis KMJC1 were introduced as probiotic capable strains. Based on the results from the current in vitro study, finding probiotics with similar or better characteristics than commercial probiotics within indigenous bacteria is quite possible. In vivo assessment of the bacteria can be considered in future studies, investigating using possibilities of these bacteria in food industries to produce functional fermented foods and in pharmaceutical industries in form of probiotic capsules.
Conflict of interest: The authors declare no conflict of interest.
- ▪ Antibacterial ▪ Adhesion assay ▪ Gastrointestinal tract ▪ Jug cheese ▪ Lactobacillus ▪ Probiotic
How to Cite
References
FAO/WHO. Guidelines for the Evaluation of Probiotics in Food. London, Ontario, Canada, April 30 and May 1, 2002.
Nejati F, Oelschlaeger TA. In Vitro characterization of Lactococcus lactis strains isolated from Iranian traditional dairy products as a potential probiotic. Appl Food Biotechnol. 2016; 3 (1): 43-51.
Buitron DI, Sepulveda S, Martinez TKM, Aguilar CN, Medina DD, Rodriguez-Herrera R, Flores-Gallegos AC. Biotech-nological approach for the production of prebiotics and search for new probiotics and their application in the food industry. Appl Food Biotechnol. 2018; 5 (4):185-192.
Nami Y, Haghshenas B, Bakhshayesh RV, Jalaly HM, Lotfi H, Eslami S, Hejazi MA. Novel autochthonous lactobacilli with probiotic aptitudes as a main starter culture for probiotic fermented milk. LWT -Food Sci Technol. 2018; 98: 85-93.
doi: 10.1016/j.lwt.2018.08.035
Melgar-Lalanne G, Rivera-Espinoza Y, Reyes Mendez AI, Hernandez-Sanchez H. In Vitro evaluation of the probiotic potential of halotolerant Lactobacilli isolated from a ripened tropical Mexican cheese. Probiotics Antimicro. 2013; 5: 239-251.
doi: 10.1007/s12602-013-9144-0.
- Jomehzadeh N, Amin M, Saki M, Hamidi H, Seyedmahmoudi M, Gorjian Z, Moghaddam M, Javaherizadeh H. Isolation and identification of potential probiotic Lactobacillus species from feces of infants in southwest Iran. Int J Infect Dis. 2020; 96:524-530.
doi: 10.1016/j.ijid.2020.05.034.
- Mahmoudi M, Khomeiri M, Saeidi M, Kashaninejad M, Davoodi H. Study of potential probiotic properties of lactic acid bacteria isolated from raw and traditional fermented camel milk. J Agric Sci Technol. 2019; 21(5): 1161-1172.
Abbasi Gaznagh M, Khosrowshahi Asl A, Bahmani M, Kian-pour F. Evaluation of nitrogen fractions during the ripening of jug cheese. J Microbiol Biotechnol. 2014; 3(4): 24-31.
Hassanzadazar H, Ehsani A. Phenotypic characterization of lactic acid bacteria isolated from traditional koopeh cheese. Global Veterinaria. 2013; 2: 148-152.
doi: 10.5829/idosi.gv.2013.10.2.6615.
Argyri AA, Zoumpopoulou G, Karatzas KAG, Tsakalidou E, Nychas GJE, Panagou E Z, Tassou CC. Selection of potential probiotic lactic acid bacteria from fermented olives by in vitro tests. Food Microbiol. 2013; 33: 282-291.
doi: 10.1016/j.fm.2012.10.005.
Jiang M, Zhang F, Wan C, Xiong Y, Shah NP, Wei H, Tao X. Evaluation of probiotic properties of Lactobacillus plantarum WLPL04 isolated from human breast milk. J Dairy Sci. 2016; 99: 1-11.
doi: 10.3168/jds.2015-10434.
CLSI. Performance standards for antimicrobial susceptibility testing; Twenty-Third Informational Supplement. 2013; Wayne, PA: Clinical and Laboratory Standards Institute.
Charteris WP, Kelly PM, Morelli L, Collins JK. Gradient diffusion antibiotic susceptibility testing of potentially probio-tic lactobacilli. J Food Protect. 2001; 64: 2007-2014.
doi: 10.4315/0362-028X-64.12.2007.
Ben Taheur F, Kouidhi B, Fdhila K, Elabed H, Ben Slama R, Mahdouani K, Bakhrouf A, Chaieb K. Anti-bacterial and anti-biofilm activity of probiotic bacteria against oral pathogens. Microb Pathogenesis. 2016; 97: 213-220.
doi: 10.1016/j.micpath.2016.06.018.
Ben Slama R, Kouidhi B, Zmantar T, Chaieb K, Bakhrouf A. Anti-listerial and anti-biofilm activities of potential probiotic Lactobacillus Strains isolated from Tunisian traditional fermented food. J Food Safety. 2013; 33: 8-16.
doi: 10.1111/jfs.12017.
Kafili T, Razavi SH, Emam Djomeh Z, Naghavi MR, Alvarez-Martin P, Mayo B. Microbial characterization of Iranian traditional Lighvan cheese over manufacturing and ripening via culturing and PCR-DGGE analysis: Identification and typing of dominant lactobacilli. Eur Food Res Technol. 2009; 229: 83-92.
doi: 10.1007/s00217-009-1028-x.
Ahmadova A, Todorov SD, Hadji-Sfaxi I, Choiset Y, Rabesona H, Messaoudi S, Kuliyev A, Franco BDGM, Chobert JM, Haertle T. Antimicrobial and antifungal activities of Lacto-bacillus curvatus strain isolated from homemade Azerbaijani cheese. Anaerobe 2013; 20: 42-49.
doi: 10.1016/j.anaerobe.2013.01.003.
Perin LM, Savo Sardaro ML, Nero LA, Neviani E, Gatti M. Bacterial ecology of artisanal Minas cheeses assessed by culture dependent and -independent methods. Food Microbiol. 2017; 65: 160-169.
doi: 10.1016/j.fm.2017.02.005.
Morales F, Morales JI, Hernandez CH, Hernandez-Sanchez H. Isolation and partial characterization of halotolerant lactic acid bacteria from two Mexican cheeses. Appl Biochem Bio-technol. 2011; 164: 889-905.
doi: 10.1007/s12010-011-9182-6.
Mahmoudi I, Ben Moussaa O, Moulouk Khaldi TE, Kebouchib M, Soligot-Hognon C, Le Roux Y, Hassouna M. Adhesion properties of probiotic Lactobacillus Strains isolated from Tunisian sheep and goat milk. J Agric Sci Technol. 2019; 21(3): 587-600.
Gomez NC, Ramiro JMP, Quecan BXV, De Melo Franco BDG. Use of potential probiotic lactic acid bacteria (LAB) biofilms for the control of Listeria monocytogenes, Salmonella Typhimurium, and Escherichia coli O157:H7 biofilms form-ation. Front Microbiol. 2016; 7: 1-15.
doi: 10.3389/fmicb.2016.00863.
Manini F, Casiraghi MC, Poutanen K, Brasca M, Erba D, Plumed-Ferrer C. Characterization of lactic acid bacteria isolated from wheat bran sourdough. LWT-Food Sci Technol. 2016; 66: 275-283.
doi: 10.1016/j.lwt.2015.10.045.
Peres CM, Alves M, Hernandez-Mendoza A, Moreira L, Silva S, Bronze MR, Vilas-Boas L, Peres C, Malcata FX. Novel isolates of lactobacilli from fermented Portuguese olive as potential probiotics. LWT-Food Sci Technol. 2014; 59: 234-246.
doi: 10.1016/j.lwt.2014.03.003.
Abriouel H, Munoz MCC, Lerma LL, Montoro BP, Bockelmann W, Pichner R, Kabisch J, Cho GS, Franz CMAP, Galvez A, Benomar N. New insights in antibiotic resistance of Lactobacillus species from fermented foods. Food Res Int. 2015; 78: 465-481.
doi: 10.1016/j.foodres.2015.09.016.
Ammor MS, Belen Florez A, Van Hoek AHAM, Reyes-Gavilan CGDL, Aarts HJM, Margolles A, Mayo B. Molecular characterization of intrinsic and acquired antibiotic resistance in lactic acid bacteria and bifidobacteria. J Mol Microbiol Biotechnol. 2008; 14: 6-15.
doi: 10.1159/000106077.
Adeniyi BA, Ayeni FA, Ogunbanwo ST. Antagonistic activities of lactic acid bacteria isolated from Nigerian fermented dairy food against organisms implicated in urinary tract infection. Biotechnol. 2006; 5: 183-188.
doi: 10.3923/biotech.2006.183.188.
Zhao S, Han J, Bie X, Lu Z, Zhang C, Lv F. Purification and characterization of plantaricin JLA-9: A novel bacteriocin against Bacillus spp. Produced by Lactobacillus plantarum JLA-9 from Suan-Tsai, a traditional Chinese fermented cabbage. J Agric Food Chem. 2016; 64: 2754-2764.
doi: 10.1021/acs.jafc.5b05717.
Zhu X, Zhao Y, Sun Y, Gu Q. Purification and characterisation of plantaricin ZJ008, a novel bacteriocin against Staphylo-coccus spp. from Lactobacillus plantarum ZJ008. Food Chem. 2014; 165: 216-223.
doi: 10.1016/j.foodchem.2014.05.034.
Corcoran BM, Stanton C, Fitzgerald GF, Ross RP. Survival of lactobacilli in acidic environments is enhanced in the presence of metabolizable sugars. Appl Environ Microbiol. 2005; 71: 3060-3067.
doi: 10.1128/AEM.71.6.3060-3067.2005.
Urdaneta V, Casadesus, J. Interactions between bacteria and bile salts in the gastrointestinal and hepatobiliary tracts. Front Med. 2017; 4: 163.
doi: 10.3389/fmed.2017.00163.
Ferrando V, Quiberoni A, Reinheimer J, Suarez V. Functional properties of Lactobacillus plantarum strains: A study In Vitro of heat stress influence. Food Microbial. 2016; 54: 154-161.
doi: 10.1016/j.fm.2015.10.003.
Thamacharoensuk T, Taweechotipatr M, Kajikawa A, Okada S, Tanasupawat S. Induction of cellular immunity interleukin-12, antiproliferative effect, and related probiotic properties of lactic acid bacteria isolated in Thailand. Ann Microbiol. 2011; 67: 511-518.
doi: 10.1007/s13213-017-1280-4.
Mahmoudi M, Khosrowshahi Asl A, Zomorodi S. The influence of probiotic bacteria on the properties of Iranian white cheese. Int J Dairy Technol. 2012; 65(4): 561-567.
doi: 10.1111/j.1471-0307.2012.00854.x
Angelescu IR, Zamfir M, Stancu MM, Grosu-Tudor SS. Identification and probiotic properties of lactobacilli isolated from two different fermented beverages. Ann Microbiol. 2019; 69: 1557-1565.
doi: 10.1007/s13213-019-01540-0.
Alp D, Kuleaşan H. Adhesion mechanisms of lactic acid bacteria: conventional and novel approaches for testing. World J Microbiol Biotechnol. 2019; 35:156.
doi: 10.1007/s11274-019-2730-x
Liu X, Liu W, Zhang Q, Tian F, Wang G, Zhang H. Screening of lactobacilli with antagonistic activity against enteroinvasive Escherichia coli. Food Control 2013; 30: 563-568.
doi: 10.1016/j.foodcont.2012.09.002.
Tuo Y, Zhang W, Zhang L, Ai L, Zhang Y, Han X, Yi H. Study of probiotic potential of four wild Lactobacillus rhamnosus strains. Anaerobe 2013; 21: 22-27.
doi: 10.1016/j.anaerobe.2013.03.007.
- Abstract Viewed: 1020 times
- pdf Downloaded: 737 times
