Isolation, Screening, Identification and Optimization of Culture Parameters to Produce γ-aminobutyric Acid by Lactiplantibacillus pentosus R13, an Isolate from Ruoc (Fermented Shrimp Paste)
Applied Food Biotechnology,
Vol. 9 No. 1 (2022),
29 December 2021
,
Page 1-8
https://doi.org/10.22037/afb.v9i1.36103
Abstract
Background and objective:
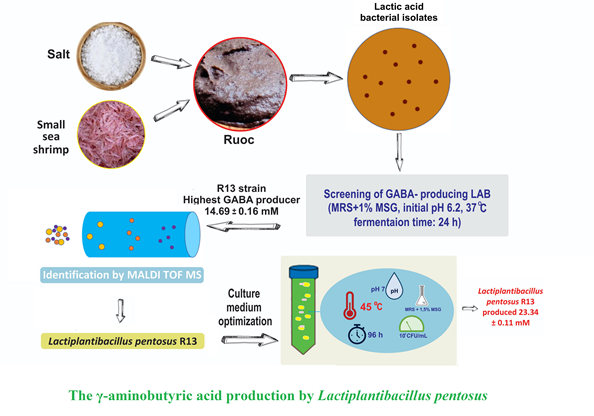
Production of γ-aminobutyric acid has recently much interested because of its benefits for health. The objective of this study was to optimize γ-aminobutyric acid production by a novel identified Lactiplantibacillus pentosus isolated from a fermented shrimp paste of ruoc.
Material and Methods:
A species of lactic acid bacterial was isolated from ‘ruoc’, a high-salt fermented shrimp paste and identified using matrix-assisted laser desorption/ionization-time of flight mass spectrometry. The γ-aminobutyric acid production was optimized using various culture conditions (initial cell density from 5 ´ 105 to 5 ´ 107 CFU ml-1, monosodium glutamate concentration of 0.5-2% (w v-1), initial pH of 4-9, incubation temperature of 30-50 °C and incubation time of 24-120 h) with one-factor-at-a-time approach.
Result and conclusion: Of 20 lactic acid bacteria isolated from ‘ruoc’, four isolates of R1, R3, R12 and R13 produced significant quantities of γ-aminobutyric acid. Isolate R13 produced the highest γ-aminobutyric acid quantity, identified as Lactiplantibacillus pentosus using matrix-assisted laser desorption/ionization-time of flight mass spectrometry. A culture media optimization study was carried out for Lactiplantibacillus pentosus R13 to improve its γ-aminobutyric acid yield. Results showed that at optimal conditions of 1.5% monosodium glutamate (w v-1), initial pH of 7, initial cell density of 5 ´ 106 CFU ml-1, cultivation temperature of 45 °C and fermentation time of 96 h, Lactiplantibacillus pentosus R13 produced 23.34 mM ±0.11 of γ-aminobutyric acid. In conclusion, γ-aminobutyric acid production by this isolate was verified to be heavily dependent on monosodium glutamate concentration, initial cell density, initial pH, incubation temperature and fermentation time.
- Fermentation
- γ-aminobutyric acid
- Lactiplantibacillus pentosus
- Lactic acid bacteria
- Optimization
How to Cite
References
Matejcekova Z, Liptakova D, Valik L. Functional probiotic products based on fermented buckwheat with Lactobacillus rhamnosus. LWT Food Sci Technol. 2017; 81: 35-41.
doi: 10.1016/j.lwt.2017.03.018
Manini F, Casiraghi MC, Poutanen K, Brasca M, Erba D, Plumed-Ferrer C. Characterization of lactic acid bacteria isolated from wheat bran sourdough. LWT Food Sci Technol. 2016; 66: 275-283.
doi: 10.1016/j.lwt.2015.10.045
Abushelaibi A, Al-Mahadin S, El-Tarabily K, Shah NP, Ayyash M. Characterization of potential probiotic lactic acid bacteria isolated from camel milk. LWT Food Sci Technol. 2017; 79: 316-325.
doi: 10.1016/j.lwt.2017.01.041
Adesulu-Dahunsi AT, Jeyaram K, Sanni AI. Probiotic and technological properties of exopolysaccharide producing lactic acid bacteria isolated from cereal-based nigerian fermented food products. Food Control. 2018; 92: 225-231.
doi: 10.1016/j.foodcont.2018.04.062
Szutowska J. Functional properties of lactic acid bacteria in fermented fruit and vegetable juices: a systematic literature review. Eur Food Res Technol. 2020; 246(3): 357-372.
doi: 10.1007/s00217-019-03425-7
Kim DH, Dasagrandhi C, Park SK, Eom SH, Huh MK, Mok JS, Kim, YM. Optimization of gamma-aminobutyric acid production using sea tangle extract by lactic acid bacterial fermentation. LWT Food Sci Technol. 2018; 90: 636-642.
doi:10.1016/j.lwt.2018.01.011
Rashmi D, Zanan R, John S, Khandagale K, Nadaf A. γ-Aminobutyric Acid (GABA): Biosynthesis, Role, Commer-cial Production and Applications. 1st ed. Vol. 57, Studies in Natural Products Chemistry. Elsevier B.V. 2018. 413-452
doi: 10.1016/B978-0-444-64057-4.00013-2
Cui Y, Miao K, Niyaphorn S, Qu X. Production of gamma-aminobutyric acid from lactic acid bacteria: A systematic review. Int J Mol Sci. 2020; 21(3): 1–21.
doi: 10.3390/ijms21030995
Diana M, Quilez J, Rafecas M. Gamma-aminobutyric acid as a bioactive compound in foods: A review. J Funct Foods. 2014; 10: 407-420.
doi: 10.1016/j.jff.2014.07.004
Shi F, Li Y. Synthesis of γ-aminobutyric acid by expressing Lactobacillus brevis-derived glutamate decarboxylase in the Corynebacterium glutamicum strain ATCC 13032. Biotechnol Lett. 2011; 33(12): 2469-2474.
doi:10.1007/s10529-011-0723-4
Lyu CJ, Liu L, Huang J, Zhao WR, Hu S, Mei LH, Yao SJ. Biosynthesis of γ-aminobutyrate by engineered Lactobacillus brevis cells immobilized in gellan gum gel beads. J Biosci Bioeng. 2019; 128(2): 123-128.
doi: 10.1016/j.jbiosc.2019.01.010
Binh TTT, Ju WT, Jung WJ, Park RD. Optimization of γ-amino butyric acid production in a newly isolated Lactobacillus brevis. Biotechnol Lett. 2014; 36(1): 93-98.
doi: 10.1007/s10529-013-1326-z
Lu X, Chen Z, Gu Z, Han Y. Isolation of γ-aminobutyric acid-producing bacteria and optimization of fermentative medium. Biochem Eng J. 2008; 41(1): 48-52.
doi: 10.1016/j.bej.2008.03.005
Peng C, Huang J, Hu S, Zhao W, Yao S, Mei L. A two-stage pH and temperature control with substrate feeding strategy for production of gamma-aminobutyric acid by Lactobacillus brevis CGMCC 1306. Chinese J Chem Eng. 2013; 21(10): 1190–1194.
doi: 10.1016/S1004-9541(13)60568-6
Cho SY, Park MJ, Kim KM, Ryu JH, Park HJ. Production of high γ-aminobutyric acid (GABA) sour kimchi using lactic acid bacteria isolated from Mukeunjee kimchi. Food Sci Biotechnol. 2011; 20(2): 403-408.
doi: 10.1007/s10068-011-0057-y
Villegas JM, Brown L, Savoy de Giori G, Hebert EM. Optimization of batch culture conditions for GABA produ-ction by Lactobacillus brevis CRL 1942, isolated from quinoa sourdough. LWT - Food Sci Technol. 2016; 67: 22-26.
doi: 10.1016/j.lwt.2015.11.027
Thuy DTB, Nguyen AT, Khoo KS, Chew KW, Cnockaert M, Vandamme P, Ho YC, Huy ND, Cocoletzi HH, Show PL. Optimization of culture conditions for gamma-aminobutyric acid production by newly identified Pediococcus pentosaceus MN12 isolated from ‘mam nem’, a fermented fish sauce. Bioengineered. 2021; 12(1): 54-62.
doi: 10.1080/21655979.2020.1857626
Dumolin C, Aerts M, Verheyde B, Schellaert S, Vandamme T, Van der Jeugt F, Canck ED, Cnockaert M, Wieme AD, Cleenwerck I, Peiren J, Dawyndt P, Vandamme P, Carlier A. Introducing SPeDE: High-Throughput dereplication and accurate determination of microbial diversity from matrix-assisted laser desorption-ionization time of flight mass spectrometry data. mSystems 2019; 4(5):1-13
doi: 10.1128/mSystems.00437-19
Nacef M, Chevalier M, Chollet S, Drider D, Flahaut C. MALDI-TOF mass spectrometry for the identification of lactic acid bacteria isolated from a French cheese: The Maroilles. Int J Food Microbiol. 2017; 247: 2-8.
doi: 10.1016/j.ijfoodmicro.2016.07.005
Siragusa S, De Angelis M, Di Cagno R, Rizzello CG, Coda R, Gobbetti M. Synthesis of γ-aminobutyric acid by lactic acid bacteria isolated from a variety of Italian cheeses. Appl Environ Microbiol. 2007; 73(22): 7283-7290.
doi:10.1128/AEM.01064-07
Doan NTL, Van Hoorde K, Cnockaert M, De Brandt E, Aerts M, Le Thanh B, Vandamme P. Validation of MALDI-TOF MS for rapid classification and identification of lactic acid bacteria, with a focus on isolates from traditional fermented foods in Northern Vietnam. Lett Appl Microbiol. 2012; 55(4):265-273.
doi:10.1111/j.1472-765X.2012.03287.x
Karaduman A, Ozaslan M, Kilic IH, Bayil-Oguzkan S, Kurt BS, Erdogan N. Identification by using MALDI-TOF mass spectrometry of lactic acid bacteria isolated from non-commercial yogurts in southern Anatolia, Turkey. Int Microbiol. 2017; 20(1): 25-30.
doi:10.2436/20.1501.01.282.
Ratanaburee A, Kantachote D, Charernjiratrakul W, Sukhoom A. Enhancement of γ-aminobutyric acid (GABA) in Nham (Thai fermented pork sausage) using starter cultures of Lactobacillus namurensis NH2 and Pediococcus pentosaceus HN8. Int J Food Microbiol. 2013; 167: 170-176.
doi: 10.1016/j.ijfoodmicro.2013.09.014
Tajabadi N, Ebrahimpour A, Baradaran A, Rahim RA, Mahyudin NA, Manap MYA, Bakar FA, Saari N. Optimization of γ-aminobutyric acid production by Lactobacillus plantarum Taj-Apis 362 from honeybees. Molecules 2015; 20(4): 6654-6669.
doi:10.3390/molecules20046654
Komatsuzaki N, Shima J, Kawamoto S, Momose H, Kimura T. Production of γ-aminobutyric acid (GABA) by Lactobacillus paracasei isolated from traditional fermented foods. Food Microbiol. 2005; 22(6): 497-504.
doi: 10.1016/j.fm.2005.01.002
Cotter PD, Hill C. Surviving the acid test: responses of gram-positive Bacteria to low pH. Microbiol Mol Biol Rev. 2003; 67(3): 429-453.
doi:10.1128/MMBR.67.3.429-453.2003
Yang SY, Lu FX, Lu ZX, Bie XM, Jiao Y, Sun LJ, Yu B. Production of γ-aminobutyric acid by Streptococcus salivarius subsp. thermophilus Y2 under submerged fermentation. Amino Acids. 2008; 34(3): 473-478.
doi: 10.1007/s00726-007-0544-x
Lin Q. Submerged fermentation of Lactobacillus rhamnosus YS9 for γ-aminobutyric acid (GABA) production. Brazilian J Microbiol. 2013; 44(1): 183-187.
doi:10.1590/S1517-83822013000100028
Sarasa SB, Mahendran R, Muthusamy G, Thankappan B, Selta DRF, Angayarkanni J. A brief review on the non-protein amino acid, gamma-amino butyric acid (GABA): Its production and role in microbes. Curr Microbiol. 2020; 77(4): 534-544.
doi: 10.1007/s00284-019-01839-w
- Abstract Viewed: 628 times
- pdf Downloaded: 566 times
