Probiotic Characterization of Cholesterol-Lowering Saccharomyces cerevisiae Isolated from Frass of Pyrrharctia isabella Caterpillars
Applied Food Biotechnology,
Vol. 8 No. 3 (2021),
15 Khordad 2021
,
Page 189-199
https://doi.org/10.22037/afb.v8i3.31729
Abstract
Background and Objectives: Cholesterol hyper-accumulation is a growing factor causing cardiovascular diseases (CVD), a leading cause of global mortality and accounts for 16.7 million deaths worldwide. As far as India is concerned, there is an increase from 25.7 to 54.7 million cases of CVD per year since the year 1990, and change in the dietary pattern being the major cause of this condition. Amongst, coronary heart disease and atherosclerosis remain prominent with its major causative agent as high cholesterol levels. However, the current treatment of these diseases is inadequate, and impose systemic toxicity such as stent thrombosis, chronic inflammation, etc., probiotics with cholesterol-lowering ability is an ideal and safe choice for the prevention of the condition.
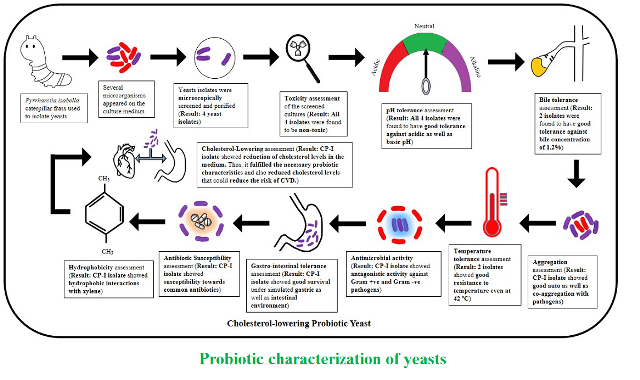
Materials and Methods: The present study involved the culture-dependent method for the enumeration of microorganisms from caterpillar frass. Upon microscopic screening, yeast isolates were further characterized for their in-vitro probiotic potential such as toxicity assessment, tolerance to pH, bile, temperature, and salt conditions, safety assessment by measuring antibiotics susceptibility, evaluating antagonistic activity, assessing survival in simulated gastrointestinal environment, measuring aggregation capability, evaluating cholesterol-lowering activity and identifying microorganisms using molecular identification tools.
Results and Conclusion: The culture-dependent approach resulted in the isolation of four yeast cultures from the frass of Pyrrharctia isabella caterpillar. Yeast cultures designated as CP-I, CP-II, CP-III, and CP-IV were resistant for bile (1.2%), wide range of pH (1.5–10), and up to 42ºC temperature along with antimicrobial activity. CP-I culture also possessed bile salt hydrolase while tolerated stressful salt conditions, gastrointestinal environment, and exhibited good aggregation properties and hydrophobicity. Interestingly, CP-I could reduce cholesterol levels by 9.16% under in-vitro conditions. Molecular identification of CP-I showed its genetic similarity 97% with Saccharomyces cerevisiae. This study demonstrated for the first-time isolation of cholesterol-lowering probiotic yeast Saccharomyces cerevisiae from caterpillar frass in in-vitro conditions.
- Caterpillar frass, cholesterol-lowering, probiotic, Pyrrharctia isabella, Saccharomyces cerevisiae
How to Cite
References
Soliman GA. Dietary cholesterol and the lack of evidence in cardiovascular disease. Nutrients 2018; 10: 780.
doi: 10.3390/nu10060780.
Pati S, Jeemon P. The changing patterns of cardiovascular diseases and their risk factors in the states of India: The global burden of disease. Lancet Glob Heal. 2018; 6.1-13.
doi:10.1016/S2214-109X(18)30407-8.
Ali K, Abo-Ali EM, Kabir MD, Riggins B, Nguy S, Li L, Srivastava U, Thinn S. A western-fed diet increases plasma HDL and LDL-cholesterol levels in apoD-/- mice. Plos One. 2014; 9:1-16.
doi:10.1371/journal.pone.0115744.
Miremadi F, Ayyash M, Sherkat F, Stojanovska L. Cholesterol reduction mechanisms and fatty acid composition of cellular membranes of probiotic Lactobacilli and Bifidobacteria. J Funct Foods. 2014; 9: 295-305. doi:10.1016/j.jff.2014.05.002.
Behera S, Pramanik K, Nayak DM. Recent advancement in the treatment of cardiovascular diseases: Conventional therapy to nanotechnology. Curr Pharm Des. 2015; 21.4479-4497
doi:10.2174/1381612821666150817104635.
FAO/WHO. Guidelines for the evaluation of probiotics in food. London, Ontario, Food and Agriculture Organization of the United Nations and World Health Organization Working Group Report. 2002; 1-11.
Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B Morelli P, Canani R, Flint H, Salminen S, Calder P, Sanders M. The international scientific association for probiotics and prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014; 11: 506-14.
doi:10.1038/nrgastro.2014.66.
Paraschiv D, Aida V, Constantin M, Ciobanu A, Bahrim GE. Study of physiological properties of some probiotics in multiple cultures with mesophilic lactic acid bacteria by Flora Danica Ch. Hansen commercial starter. Ann Univ Dunarea Jos Galati VI- Food Technol. 2010; 35: 56-65.
Das B, Ghosh TS, Kedia S, Rampal R, Saxena S, Bag S, Mitra R, Dayal M, Mehta O, Surendranath A, Travis S, Tripathi P, Nair G and Ahuja V. Analysis of the gut microbiome of rural and urban healthy Indians living in sea level and high altitude areas. Sci Rep. 2018; 8: 1-14.
doi:10.1038/s41598-018-28550-3.
Dudek-Wicher RK, Junka A, Bartoszewicz M. The influence of antibiotics and dietary components on gut microbiota. Prz Gastroenterol. 2018; 13: 85-92.
doi:10.5114/pg.2018.76005.
Ganguly N, Bhattacharya S, Sesikeran B, Nair G, Ramakrishna B, Sachdev H, Batish V, Kanagaspathy A, Muthuswamy V, Kathuria S, Katoch V, Satyanarayana K, Toteja G, Rahi M, Bhan M, Kapur R, Hemlatha R. ICMR-DBT guidelines for evaluation of probiotics in food. Indian Med Res. 2011; 134(1): 22-25.
Guarner F, Khan A, Garisch J, Eliakim R, Gangl A, Thomson A. World gastroenterology organisation global guidelines. J Clin Gastroenterol. 2012; 46: 468-81.
doi:10.1097/MCG.0b013e3182549092.
Nayak S. Biology of eukaryotic probiotics. Probiotics 2011;21: 29-55.
doi: 10.1007/978-3-642-20838-6_2
Engel P, Moran NA. The gut microbiota of insects - diversity in structure and function. FEMS Microbiol Rev. 2013; 37: 699-735.
doi:10.1111/1574-6976.12025.
Sonavale R, Kunchiraman B. Probiotic characteristics of anti-candida Bacillus tequilensis isolated from sheep milk and buffalo colostrums. Int J Heal Sci Res. 2018; 8: 254-260.
Chanthala L, Appaiah A. In vitro probiotic characterization of yeasts of food and environmental origin. Int J Probiotics Prebiotics. 2014; 9: 87-92.
Khisti U, Kathade S, Aswani M, Anand P, Kunchirman B. Isolation and identification of Saccharomyces cerevisiae from caterpillar frass and their probiotic characterization. Biosci Biotechnol Res Asia. 2019; 16: 179-186. doi:10.13005/bbra/2735.
Ogunremi OR, Sanni AI, Agrawal R. Probiotic potentials of yeasts isolated from some cereal-based nigerian traditional fermented food products. J Appl Microbiol. 2015; 119: 797-808.
doi:10.1111/jam.12875.
Chelliah R, Ramakrishnan SR, Prabhu PR, Antony U. Evaluation of antimicrobial activity and probiotic properties of wild-strain Pichia kudriavzevii isolated from frozen idli batter. Yeast. 2016; 33: 385-401.
doi:10.1002/yea.3181.
Palande V, Meora R, Sonavale R, Makashir M, Modak M, Kapse N, Dhakephalkar P, Ranjekar P, Kunchiraman B. Inhibition of pathogenic strains of Candida albicans and non-albicans by Bacillus species isolated from traditional Indian fermented food preparations. Int J Curr Microbiol App Sci. 2015; 4: 691-699.
Charteris K, Morelli C. Development and application of an in vitro methodology to determine the transit tolerance of potentially probiotic Lactobacillus and Bifidobacterium species in the upper human gastrointestinal tract. J Appl Microbiol. 1998; 84: 759-768.
doi:10.1046/j.1365-2672.1998.00407.x.
Chandran HC, Keerthi TR. Probiotic potency of Lactobacillus plantarum KX519413 and KX519414 isolated from honey bee gut. FEMS Microbiol Lett. 2018; 365.
doi:10.1093/femsle/fnx285.
Bauer AW, Perry DM, Kirby WM. Single-disk antibiotic-sensitivity testing of Staphylococci; an analysis of technique and results. AMA Arch Intern Med. 1959; 104: 208-216.
doi:10.1001/archinte.1959.00270080034004.
Zheng Y, Lu Y, Wang J, Young L, Pan C, Huang Y. Probiotic Properties of Lactobacillus strains isolated from Tibetan kefir grains. Plos One. 2013; 8: 1-8.
doi:10.1371/journal.pone.0069868.
Polkade AV, Ramana VV, Joshi A, Pardesi L, Shouche YS. Rufibacter immobilis sp. nov., isolated from a high-altitude saline lake. Int J Syst Evol Microbiol. 2015; 65: 1592-1597.
doi:10.1099/ijs.0.000144.
Billah MM, Hoque MZ, FA, Hossain KM, MSM R, Islam KMD. Isolation, identification and analysis of probiotic properties of Lactobacillus spp. from selective regional yoghurts. World J Dairy Food Sci. 2010; 5: 39-46.
Shehata MG, El Sohaimy SA, El-Sahn MA, Youssef MM. Screening of isolated potential probiotic lactic acid bacteria for cholesterol lowering property and bile salt hydrolase activity. Ann Agric Sci. 2016; 61: 65-75. doi:10.1016/j.aoas.2016.03.001.
Gotcheva V, Hristozova E, Hristozova T, Guo M, Roshkova Z, Angelov A. Assessment of potential probiotic properties of lactic acid bacteria and yeast strains. Food Biotechnol. 2002; 16: 211-225.
doi:10.1081/FBT-120016668.
Toit MD, Dicks LMT, Holzapfel WH. Identification of heterofermentative Lactobacilli isolated from pig faeces by numerical analysis of total soluble cell protein patterns and RAPD-PCR. Lett Appl Microbiol. 2003; 37: 12-16. doi:10.1046/j.1472-765X.2003.01334.x.
Wu Q, Shah N. Effects of elaidic acid, a predominant industrial trans fatty acid, on bacterial growth and cell surface hydrophobicity of Lactobacilli. J Food Sci. 2014; 79: 2485-2490.
doi:10.1111/1750-3841.12695.
Krausova G, Hyrslova I, Hynstova I. In Vitro evaluation of adhesion capacity, hydrophobicity and auto-aggregation of newly isolated potential probiotic strains. Fermentation. 2019; 5: 100.
doi:10.3390/fermentation5040100.
Reynolds J, Moyes RB, Breakwell DP. Differential staining of bacteria: Acid fast stain. Curr Protoc Microbiol. 2009; 15: 31- 35.
doi:10.1002/9780471729259.mca03hs15.
Viswanathan S, Preethi G, Veilumuthu P, Rajesh M, Suba P. Probiotic studies in colostrum of buffalo. Glob Vet. 2015; 14: 199-204.
doi: 10.5829/idosi.gv.2015.14.02.92179
Hickson M. Probiotics in the prevention of antibiotic-associated diarrhoea and Clostridium difficile infection. Therap Adv Gastroenterol. 2011; 4: 185-197. doi:10.1177/1756283X11399115.
Kathade SA, Aswani MA, Anand PK, Jagtap S, Bipinraj NK. Isolation of Lactobacillus from donkey dung and its probiotic characterization. Korean J Microbiol. 2020; 56(2): 160-169. doi:10.7845/kjm.2020.0038.
Yang Y, Liu Y, Zhou S, Huang L, Chen Y, Huan H. Bile salt hydrolase can improve Lactobacillus plantarum survival in gastrointestinal tract by enhancing their adhesion ability. FEMS Microbiol Lett. 2019; 366: 1-8.
doi:10.1093/femsle/fnz100.
Pereira DIA, Gibson GR. Cholesterol assimilation by lactic acid bacteria and Bifidobacteria isolated from the human gut. Appl Environ Microbiol. 2002; 68: 4689-4693.
doi:10.1128/aem.68.9.4689-4693.2002.
Liong MT, Shah NP. Bile salt deconjugation ability, bile salt hydrolase activity and cholesterol co-precipitation ability of Lactobacilli strains. Int Dairy J. 2005; 15:391-398.
doi:10.1016/j.idairyj.2004.08.007.
Kumar M, Nagpal R, Kumar R. Cholesterol-lowering probiotics as potential biotherapeutics for metabolic diseases. Exp Diabetes Res. 2012; 2012: 902-917.
doi: 10.1155/2012/902917
- Abstract Viewed: 677 times
- pdf Downloaded: 591 times
