In vitro Antioxidant and α-Glucosidase Inhibitory Activities of Lactobacillus spp. Isolated from Indonesian Kefir Grains
Applied Food Biotechnology,
Vol. 8 No. 1 (2021),
14 December 2020
,
Page 39-46
https://doi.org/10.22037/afb.v8i1.30367
Abstract
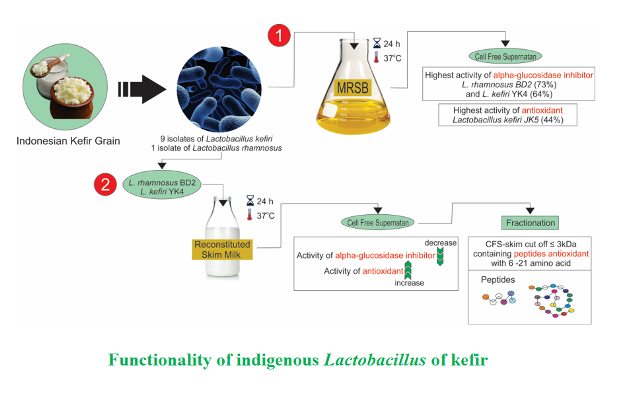
Background and Objective: In this study, nine Lactobacillus kefiri and one Lactobacillus rhamnosus isolates with kefir grain origin have been demonstrated to include potentials as probiotics. The aim of this study was to investigate ability of the isolates to produce compounds with α-glucosidase inhibitory and antioxidant activities and identify peptides with MW of ≤ 3 kDa in cell-free supernatants.
Material and Methods: All isolates were cultured in de Man, Rogosa and Sharpe broth media at 37 oC for 24 h. Assessment of α-glucosidase inhibitory and antioxidant activities was carried out on cell-free supernatants. Assessment of optimum incubation time was carried out on two isolates with the highest α-glucosidase inhibitory and antioxidant activities. The two isolates were used to ferment reconstituted skim milk. Cell-free supernatant of the fermented skim milk was fractionated using filters of 10 and 3 kDa. Then, peptides in fractions of ≤ 3 kDa were identified.
Results and Conclusion: The highest α-glucosidase inhibitory activity was seen in Lactobacillus rhamnosus BD2 and Lactobacillus kefiri YK4 as 73.58 and 64.31%, respectively. The highest antioxidant activity was observed in Lactobacillus kefiri JK5 and Lactobacillus kefiri JK17 as 44.31 and 41.57%, respectively. When Lactobacillus rhamnosus BD2 and Lactobacillus kefiri YK4 were cultured in reconstituted skim milk, their α-glucosidase inhibitory activities respectively decreased to 25.72 and 36.16% while the antioxidant activities respectively increased to 74.53 and 80.92%. Fractionation of the cell-free supernatants from fermented reconstituted skim milk of Lactobacillus kefiri YK4 showed that the highest antioxidant activity was included in fractions greater than 10 kDa. Although fractions of 3 kDa or less exhibited quite high antioxidant activities. Identification of peptides in fractions of 3 kDa or less showed that the peptides were mostly derived form β-casein. Of these peptides, two peptides with sequences of FPPQSV and YQEPVLGPVRGPFPIIV have been reported to include antioxidant activities.
Conflict of interest: The authors declare no conflict of interest.
- ▪ Lactobacillus spp ▪ Kefir grains ▪ Peptides ▪ α-glucosidase inhibitors ▪ Antioxidants
How to Cite
References
Maria IC, Molyneaux L, Limacher-Gisler F, Al-Saeed A, Luo C, Wu T, Twigg SM, Yue DK, Wong J. Long-term complications and mortality in young-onset diabetes. Diabetes Care 2013; 36:3863-3869.
doi: 10.2337/dc12-2455.
Chatterjee S, Khunti K, Davies MJ. Type 2 diabetes.The Lancet 2017; 389: 2239-2251.
doi: 10.1016/S0140-6736(17)30058-2.
Liu Z, Ma S. Recent Advances in Synthetic α-Glucosidase Inhibitors. Chem Med Chem 2017; 12: 819-829.
doi: 10.1002/cmdc.201700216.
Pisoschi AM, Pop A. The role of antioxidants in the chemistry of oxidative stress: A review. Eur J Med Chem. 2015; 97: 55-74.
doi: 10.1016/j.ejmech.2015.04.040.
Wang Y, Wu Y, Wang Y, Xu H, Mei X, Yu D, Wang Y, Li W. Antioxidant properties of probiotic bacteria. Nutrients 2017; 9: 512.
doi: 10.3390/nu9050521.
Chen P, Zhang Q, Dang H, Liu X, Tian F, Zhao J, Chen Y, Zhang H, Chen W. Screening for potential new probiotic based on probiotic properties and α-glucosidase inhibitory activity. Food Control 2014; 35: 65-72.
doi: 10.1016/j.foodcont.2013.06.027.
Panwar H, Calderwood D, Grant IR, Grover S, Green BD. Lactobacillus strains isolated from infant faeces possess potent inhibitory activity against intestinal alpha-and beta-glucosidases suggesting anti-diabetic potential. Eur J Nutr. 2014; 53: 1465-1474.
doi: 10.1007/s00394-013-0649-9.
Muganga L, Liu X, Tian F, Zhao J, Zhang H, Chen W. Screening for lactic acid bacteria based on antihyperglycaemic and probiotic potential and application in synbiotic set yoghurt. J Funct Foods. 2015; 16: 125-136.
doi: 10.1016/j.jff.2015.04.030.
Zeng Z, Luo J, Zuo F, Zhang Y, Ma H, Chen S. Screening for potential novel probiotic Lactobacillus strains based on high dipeptidyl peptidase IV and α-glucosidase inhibitory activity. J Funct Foods. 2016; 20: 486-495.
doi: 10.1016/j.jff.2015.11.030.
Son SH, Jeon HL, Yang SJ, Lee NK, Paik HD. In vitro characterization of Lactobacillus brevis KU15006, an isolate from kimchi, reveals anti-adhesion activity against foodborne pathogens and antidiabetic properties. Microb Pathog. 2017; 112: 135-141.
doi: 10.1016/j.micpath.2017.09.053.
Rossi M, Amaretti A, Raimondi S. Folate production by probiotic bacteria. Nutrients 2011; 3: 118-134.
doi: 10.3390/nu3010118.
Ejtahed HS, Mohtadi-Nia J, Homayouni-Rad A, Niafar M, Asghari-Jafarabadi M, Mofid V. Probiotic yogurt improves antioxidant status in type 2 diabetic patients. Nutrition 2012; 28: 539-543.
doi: 10.1016/j.nut.2011.08.013.
LeBlanc JG, Chain F, Martin R, Bermunez-Humaran LG, Courau S, Langella P. Beneficial effects on host energy metabolism of short-chain fatty acids and vitamins produced by commensal and probiotic bacteria. Microb Cell Fact. 2017; 16: 1-10.
doi: 10.1186/s12934-017-0691-z.
Yusuf D, Nuraida L, Dewanti-Hariyadi R, Hunaefi D. In Vitro characterization of lactic acid bacteria from Indonesian kefir grains as probiotics with cholesterol-lowering effect. J Microbiol Biotechnol. 2020; 30: 726-732.
doi: 10.4014/jmb.1910.10028.
Farida E, Jenie BSL, Nuraida L, Giriwono PE. Aktivitas antioksidan dan penghambatan α-glukosidase oleh ekstrak etanol bakteri asam laktat indigenus. J Teknol dan Ind Pangan 2019; 30: 56-63.
doi: 10.6066/jtip.2019.30.1.56.
Yang E, Fan L, Yan J, Jiang Y, Doucette C, Fillmore S, Walker B. Influence of culture media, pH and temperature on growth and bacteriocin production of bacteriocinogenic lactic acid bacteria. AMB Express 2018; 8: 10.
doi: 10.1186/s13568-018-0536-0.
Church FC, Swaisgood HE, Porter DH, Catignani GL. Spectrophotometric assay using o-phthaldialdehyde for determination of proteolysis in milk and isolated milk proteins. J Dairy Sci. 1983; 66: 1219-1227.
doi: 10.3168/jds.S0022-0302(83)81926-2.
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951; 193: 265-275.
Daliri EBM, Lee BH, Kim JH, Oh DH. Novel angiotensin I-converting enzyme inhibitory peptides from soybean protein isolates fermented by Pediococcus pentosaceus SDL1409. LWT. 2018; 93: 88-93.
doi: 10.1016/j.lwt.2018.03.026.
Nguyen VB, Wang SL. New novel α-glucosidase inhibitors produced by microbial conversion. Process Biochem. 2018; 65: 228-232.
doi: 10.1016/j.procbio.2017.11.016.
Ramchandran L, Shah NP. Proteolytic profiles and angiotensin-I converting enzyme and α-glucosidase inhibitory activities of selected lactic acid bacteria. J Food Sci. 2008; 73: 75-81.
doi: 10.1111/j.1750-3841.2007.00643.x.
Adesulu-Dahunsi AT, Jeyaram K, Sanni AI. Production of exopolysaccharide by strains of Lactobacillus plantarum YO175 and OF101 isolated from traditional fermented cereal beverage. PeerJ. 2018; 6: e5326.
doi: 10.7287/peerj.preprints.26579v1.
Kullisaar T, Songisepp E, Aunapuu M, Kilk K, Arend A, Mikelsaar M, Rehema A, Zilmer M. Complete glutathione system in probiotic Lactobacillus fermentum ME-3. Appl Biochem Microbiol. 2010; 46: 481-486.
doi: 10.1134/S0003683810050030.
Ramesh V, Kumar R, Singh RRB, Kaushik JK, Mann B. Comparative evaluation of selected strains of lactobacilli for the development of antioxidant activity in milk. Dairy Sci Technol. 2012; 92: 179-188.
doi: 10.1007/s13594-011-0048-z.
Matejcekova Z, Liptakova D, Spodniakova S, Valik L. Characterization of the growth of Lactobacillus plantarum in milk in dependence on temperature. Acta Chim Slovaca. 2016; 9: 104-108.
doi: 10.1515/acs-2016-0018.
Sabeena Farvin KH, Baron CP, Nielsen NS, Otte J, Jacobsen C. Antioxidant activity of yoghurt peptides: Part 2-Characterisation of peptide fractions. Food Chem. 2010; 123: 1090-1097.
doi: 10.1016/j.foodchem.2010.05.029.
Castro RJS, Sato HH. Biologically active peptides: Processes for their generation, purification and identification and applications as natural additives in the food and pharmaceutical industries. Food Res Int. 2015; 74: 185-198.
doi: 10.1016/j.foodres.2015.05.013.
Malaguti M, Dinelli G, Leoncini E, Bregola V, Bosi S, Cicero AFG, Hrelia S. Bioactive peptides in cereals and legumes: Agronomical, biochemical and clinical aspects. Int J Mol Sci. 2014; 15: 21120-21135.
doi: 10.3390/ijms151121120.
Hafeez Z, Cakir-Kiefer C, Roux E, Perrin C, Miclo L, Dary-Mourot A. Strategies of producing bioactive peptides from milk proteins to functionalize fermented milk products. Food Res Int. 2014; 63: 71-80.
doi: 10.1016/j.foodres.2014.06.002.
Teneva-Angelova T, Hristova I, Pavlov A, Beshkova D. Lactic Acid Bacteria-From Nature Through Food to Health. In: Advances in Biotechnology for Food Industry. Elsevier, 2018: pp. 91-133.
doi: 10.1016/B978-0-12-811443-8.00004-9.
Zhou M, Gucinski AC, Boyne MT. Performance metrics for evaluating system suitability in liquid chromatography-mass spectrometry peptide mass mapping of protein therapeutics and monoclonal antibodies. MAbs. 2015; 7: 1104-1117.
doi: 10.1080/19420862.2015.1074364.
Conway V, Gauthier SF, Pouliot Y. Antioxidant activities of buttermilk proteins, whey proteins, and their enzymatic hydrolysates. J Agric Food Chem. 2013; 61: 364-372.
doi: 10.1021/jf304309g.
Sandree C, Gleizes A, Forestier F, Gorges-Kergot R, Chilmonczyk S, Leonil J, Moreau MC, Labarre C. A peptide derived from bovine β-casein modulates functional properties of bone marrow-derived macrophages from germfree and human flora-associated mice. J Nutr. 2001; 131: 2936-2942.
doi: 10.1093/jn/131.11.2936.
Eisele T, Stressler T, Kranz B, Fischer L. Bioactive peptides generated in an enzyme membrane reactor using Bacillus lentus alkaline peptidase. Eur Food Res Technol. 2013; 236: 483-490.
doi: 10.1007/s00217-012-1894-5.
Sonklin C, Laohakunjit N, Kerdchoechuen O. Assessment of antioxidant properties of membrane ultrafiltration peptides from mungbean meal protein hydrolysates. PeerJ. 2018; 6: e5337
doi: 10.7717/peerj.5337.
- Abstract Viewed: 986 times
- pdf Downloaded: 817 times
