A Kinetic Modeling of Growth and Mycelial Exopolysaccharide Production by Lentinus edodes (Shiitake Edible Mushroom) Enhancemet of production in volatile co-cultures for shiitake edible mushroom
Applied Food Biotechnology,
Vol. 9 No. 1 (2022),
29 Dey 2021
,
Page 67-78
https://doi.org/10.22037/afb.v9i1.36579
Abstract
Background and Aims:
Lentinus edodes (Shiitake) is a rich source of secondary metabolites, including exopolysaccharides. These compounds strengthen the immune system and play essential roles in prevention and treatment of several diseases, including cancers. A way to increase production of polysaccharides is the use of elicitors. Examples of these elicitors include microbial volatile organic compounds, which are produced in microo-rganism co-cultures. The objective of this study was to investigate effects of these compounds on production of Shiitake exopolysaccharides.
Materials and Methods:
To decrease cultivation time, Shiitake was cultured in four culture media, including (1) potato dextrose broth, (2) potato dextrose broth and D-glucose, (3) malt extract broth and (4) malt extract broth and D-glucose. After selecting appropriate culture media, fungal growth curve, kinetic growth of pellets and filamentous morphology were studied. Novel method of simultaneous aerial co-culture was used to increase production of Shiitake exopolysaccharides, which acted as an elicitor by inducing microbial volatile organic compounds of other microorganisms. Microbial volatile organic compounds were analyzed using gas chromatography-mass spectroscopy.
Results:
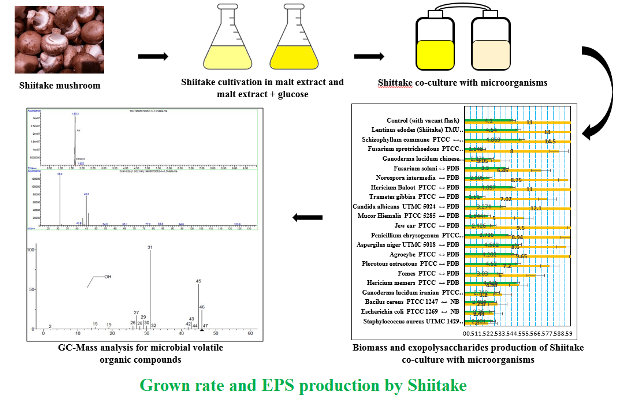
Malt extract medium containing glucose was selected for submerged and solid cultures of Shiitake and the growth time decreased to 18 d. Shiitake biomass production included 11 g.l-1. Filamentous morphology included higher production rates due to higher surface-to-volume ratios, compared to that the pellet morphology did. Shiitake fungal biomass and exopolysaccharides in co-cultures with Aspergillus niger included 14 and 4 g.l-1, respectively. Furthermore, biomass and exopolysaccharides included 11 and 4.7 g.l-1 in co-cultures with Schizophyllum commune, respectively. Microbial volatile organic compounds produced by Aspergillus niger and Schizophyllum commune in co-cultures, as elicitors, increased biomass and exopolysaccharide productions in Shiitake. Therefore, it suggests that microorganism co-cultivation is a low-cost effective method for Shiitake exopolysaccharide production.
- Shiitake, Aerial co-culture, Microbial volatile organic compounds, Lentinan, Exopolysaccharide
How to Cite
References
Luo D, Wu J, Ma Z, Tang P, Liao X, Lao F. Production of high sensory quality Shiitake mushroom (Lentinus edodes) by pulsed air-impingement jet drying (AID) technique. Food Chem. 2021; 341:128290.
doi:10.1016/j.foodchem.2020.128290
Hobbs C. Medicinal value of Lentinus edodes (Berk.) Sing.(Agaricomycetideae). A literature review. Int J Med Mushrooms. 2000; 2(4).
doi:10.1615/IntJMedMushr.v2.i4.90.
Sheng K, Wang C, Chen B, Kang M, Wang M, Liu K,Wang M. Recent advances in polysaccharides from Lentinus edodes (Berk.): Isolation, structures and bioactivities. Food Chem. 2021; 129883.
doi:10.1016/j.foodchem.2021.129883.
Reshetnikov SV,Tan K-K. Higher Basidiomycota as a source of antitumor and immunostimulating polysaccharides. Int J Med Mushrooms. 2001; 3(4).
doi:10.1615/IntJMedMushr.v3.i4.80.
Alvandi H, Hatamian-Zarmi A, Ebrahimi Hosseinzadeh B, Mokhtari-Hosseini Z-B. Optimization of production conditions for bioactive polysaccharides from fomes fomen-tarius and investigation of antibacterial and antitumor activities. Iran J Med Microbiol. 2020; 14(6): 596-611.
doi:10.30699/ijmm.14.6.596
Alvandi H, Hatamian-Zarmi A, Ebrahimi Hosseinzadeh B, Mokhtari-Hosseini Z-B, Aghajani H. Effect of production, extraction and purification methods on anti-cancer property of fungal polysaccharides. Dev Biol. 2020; 12(1): 27-42.
Alvandi H, Hatamian-Zarmi A, Hoseinzadeh BE, Mokhtari-Hosseini ZB, Langer E, Aghajani H. Improving the biological properties of fomes fomentarius MG835861 exopolysacch-aride by bioincorporating selenium into its structure. Carbohydr Polym Technol Appl. 2021; 100159.
doi:10.1016/j.carpta.2021.100159.
Yasrebi N, Hatamian Zarmi AS, Larypoor M. Optimization of chitosan production from Iranian medicinal fungus trametes-versicolor by Taguchi method and evaluation of antibacterial properties. Iran J Med Microbiol. 2020; 14(3): 186-200.
doi:10.30699/ijmm.14.3.186.
Zeynali M, Hatamian-Zarmi A, Larypoor M. Evaluation of chitin-glucan complex production in submerged culture of medicinal mushroom of Schizophilum commune: Optimiz-ation and growth kinetic. Iran J Med Microbiol. 2019; 13(5): 406-424.
doi:10.30699/ijmm.13.5.406.
Nojoki F, Hatamian-Zarmi A, Hosseinzadeh BE, Mir-derikvand M. Investigation and optimization effects of ultrasound waves to produce Ganoderic acid, anti-cancer mushrooms metabol-ite. Iran J Med Microbiol. 2017; 11(1): 58-66.
Nojoki F, Hatamian-Zarmi A, Mir-Drikvand M, Ebrahimi-Hosseinzadeh B, Mokhtari-Hosseini Z-b, Kalantari-Dehaghi S, Esmaeilifar M. Impact of rifampin induction on the fermentation production of Ganoderic acids by medicinal mushroom Ganoderma lucidum. Appl Food Biotechnol. 2016; 3(2): 91-98.
doi:10.22037/afb.v3i2.10797
Heydarian M, Hatamian ZA, Amoabediny G, Yazdian F, Doryab A. Synergistic effect of elicitors in enhancement of ganoderic acid production: Optimization and gene expression studies. Appl Food Biotechnol. 2015.
doi:10.22037/afb.v2i3.8715
Esmaelifar M, Hatamian-Zarmi A, Alvandi H, Azizi M, Mokhtari-Hosseini ZB, Ebrahimi-Hoseinzadeh B. Optimiz-ation of antioxidant activities and intracellular polysaccharide contents using Agaricus bisporus extract as elicitor in submerged fermenting Ganoderma lucidum. Appl Food Biotechnol. 2021; 8(4): 297-306.
doi:10.22037/afb.v8i4.35155
Ben Salah I, Aghrouss S, Douira A, Aissam S, El Alaoui-Talibi Z, Filali-Maltouf A, El Modafar C. Seaweed polysaccharides as bio-elicitors of natural defenses in olive trees against verticillium wilt of olive. J Plant Interact. 2018; 13(1): 248-255.
doi:10.1080/17429145.2018.1471528.
Alcantara J, Mondala A. Optimization of slurry fermentation for succinic acid production by fungal co-culture. Chem Eng Trans. 2021; 86:1525-1530.
doi:10.3303/CET2186255
Leng L, Li W, Chen J, Leng S, Chen J, Wei L, Peng H, Li J, Zhou W, Huang H. Co-culture of fungi-microalgae consortium for wastewater treatment: A review. Bioresour Technol. 2021; 330:125008.
doi:10.1016/j.biortech.2021.125008.
Peng X-Y, Wu J-T, Shao C-L, Li Z-Y, Chen M,Wang C-Y. Co-culture:Stimulate the metabolic potential and explore the molecular diversity of natural products from microorganisms. Mar Life Sci Technol. 2021;1-12.
doi:10.1007/s42995-020-00077-5
Kalantari-Dehaghi S, Hatamian-Zarmi A, Ebrahimi-Hosseinzadeh B, Mokhtari-Hosseini Z-B, Nojoki F, Hamedi J, Hosseinkhani S. Effects of microbial volatile organic compounds on Ganoderma lucidum growth and Ganoderic acids production in co-v-cultures (volatile co-cultures). Prep Biochem Biotechnol. 2019; 49(3): 286-297.
doi:10.1080/10826068.2018.1541809.
Asadi F, Barshan-Tashnizi M, Hatamian-Zarmi A, Davoodi-Dehaghani F, Ebrahimi-Hosseinzadeh B. Enhancement of exopolysaccharide production from Ganoderma lucidum using a novel submerged volatile co-culture system. Fungal Biol. 2021; 125(1): 25-31.
doi:10.1016/j.funbio.2020.09.010.
Garbeva P, Hordijk C, Gerards S, de Boer W. Volatile-mediated interactions between phylogenetically different soil bacteria. Front Microbiol. 2014; 5: 289.
doi:10.3389/fmicb.2014.00289.
Venkataraman A, Rosenbaum MA, Werner JJ, Winans SC, Angenent LT. Metabolite transfer with the fermentation product 2, 3-butanediol enhances virulence by Pseudomonas aeruginosa. ISME J. 2014; 8(6): 1210-1220.
doi:10.1038/ismej.2013.232.
Savoie J-M, Mata G, Billette C. Extracellular laccase production during hyphal interactions between Trichoderma sp. and Shiitake, Lentinula edodes. Appl Microbiol Biotechnol. 1998; 49(5): 589-593.
doi:10.1007/s002530051218.
Netzker T, Fischer J, Weber J, Mattern DJ, Konig CC, Valiante V, Schroeckh V, Brakhage AA. Microbial communication leading to the activation of silent fungal secondary metabolite gene clusters. Front Microbiol. 2015; 6: 299.
doi:10.3389/fmicb.2015.00299.
Sharma V, Kumar S, Kumar R, Singh R,Verma D. Cultural requirements, enzyme activity profile, molecular identity and yield potential of some potential strains of Shiitake (Lentinula edodes). Mushroom Res. 2013; 22: 105-110.
Feng Y-L, Li W-Q, Wu X-Q, Cheng J-W, Ma S-Y. Statistical optimization of media for mycelial growth and exo-polysaccharide production by Lentinus edodes and a kinetic model study of two growth morphologies. Biochem Eng J. 2010; 49(1): 104-112.
doi:10.1016/j.bej.2009.12.002.
Feng J, Zhang J-S, Jia W, Yang Y, Liu F, Lin C-C. An unstructured kinetic model for the improvement of triterpenes production by Ganoderma lucidum G0119 based on nitrogen source effect. Biotechnol Bioproce Eng. 2014; 19(4): 727-732.
Schalchli H, Hormazabal E, Becerra J, Birkett M, Alvear M, Vidal J,Quiroz A. Antifungal activity of volatile metabolites emitted by mycelial cultures of saprophytic fungi. J Chem Ecol. 2011; 27(6): 503-513.
doi: 10.1080/02757540.2011.596832
Fan X, Jiao X, Liu J, Jia M, Blanchard C, Zhou Z. Characterizing the volatile compounds of different sorghum cultivars by both GC-MS and HS-GC-IMS. Food Res Int. 2021; 140: 109975.
doi:10.1016/j.foodres.2020.109975
Hiroko Hassegawa R, Megumi Kasuya MC, Dantas Vanetti MC. Growth and antibacterial activity of Lentinula edodes in liquid media supplemented with agricultural wastes. Electron J Biotechnol. 2005; 8(2): 94-99.
doi:10.4067/S0717-34582005000200011.
Elisashvili VI, Tan K-K, Chichua D, Kachlishvili E. Extracellular polysaccharide production by culinary-medicinal Shiitake mushroom Lentinus edodes (Berk.) Singer and Pleurotus (Fr.) P. Karst. species depending on carbon and nitrogen source. Int J Med Mushrooms. 2004; 6(2).
doi:10.1615/IntJMedMushr.v6.i2.70.
Enman J, Hodge D, Berglund KA, Rova U. Production of the bioactive compound eritadenine by submerged cultivation of shiitake (Lentinus edodes) mycelia. J Agric Food Chem. 2008; 56(8): 2609-2612.
doi:10.1021/jf800091a.
Toyotome T, Takino M, Takaya M, Yahiro M, Kamei K. Identification of volatile sulfur compounds produced by Schizophyllum commune. J Fungus. 2021; 7(6): 465.
doi:10.3390/jof7060465.
Seesaard T, Thippakorn C, Kerdcharoen T, Kladsomboon SA. Hybrid electronic nose system for discrimination of pathogenic bacterial volatile compounds. Anal Methods. 2020; 12(47): 5671-5683.
doi:10.1039/d0ay01255f.
Jaddoa HH, Hameed IH, Mohammed GJ. Analysis of volatile metabolites released by Staphylococcus aureus using Gas chromatography-Mass spectrometry and determination of its antifungal activity. Orient J Chem. 2016; 32(4): 8-24.
doi:10.13005/ojc/320439.
Filipiak W, Sponring A, Baur MM, Filipiak A, Ager C, Wiesenhofer H, Nagl M, Troppmair J, Amann A. Molecular analysis of volatile metabolites released specifically by staphylococcus aureus and pseudomonas aeruginosa. BMC Microbiol. 2012; 12(1): 1-16.
doi:10.1186/1471-2180-12-113.
Siddiquee S, Al Azad S, Bakar FA, Naher L, Kumar SV. Separation and identification of hydrocarbons and other volatile compounds from cultures of Aspergillus niger by GC-MS using two different capillary columns and solvents. J Saudi Chem Soc. 2015; 19(3): 243-256.
doi:10.1016/j.jscs.2012.02.007.
Li P, Wang H, Liu G, Li X,Yao J. The effect of carbon source succession on laccase activity in the co-culture process of Ganoderma lucidum and a yeast. Enzyme Microb Technol. 2011; 48(1): 1-6.
doi:10.1016/j.enzmictec.2010.07.005.
Antonelli M, Donelli D, Barbieri G, Valussi M, Maggini V, Firenzuoli F. Forest volatile organic compounds and their effects on human health: A state-of-the-art review. Int J Environ Res Public Health. 2020; 17(18): 6506.
doi:10.3390/ijerph17186506
Thorn RMS,Greenman J. Microbial volatile compounds in health and disease conditions. J Breath Res. 2012; 6(2): 024001.
doi:10.1088/1752-7155/6/2/024001
McSweeney C, Denman S. Effect of sulfur supplements on cellulolytic rumen micro‐organisms and microbial protein synthesis in cattle fed a high fibre diet. J Appl Microbiol. 2007; 103(5): 1757-1765.
doi: 10.1111/j.1365-2672.2007.03408.x.
- Abstract Viewed: 515 times
- pdf Downloaded: 506 times
