Anti-Diabetic Properties of Hydrolysates from Egg White Proteins Using Immobilized Enzymes Followed by in vitro Gastrointestinal Digestion
Applied Food Biotechnology,
Vol. 7 No. 4 (2020),
18 August 2020
,
Page 235-249
https://doi.org/10.22037/afb.v7i4.30219
Abstract
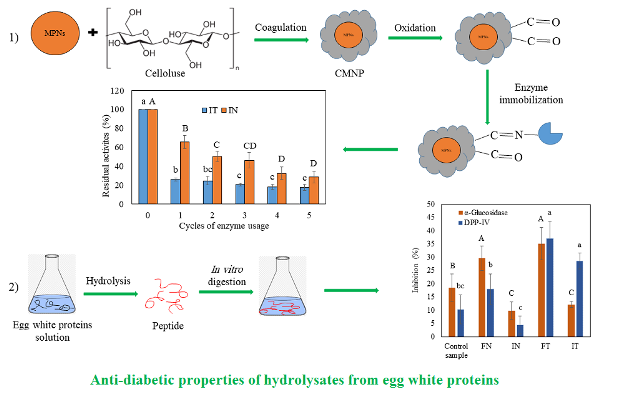
Background and Objective: Enzyme-assisted hydrolysis of the proteins from food sources is an effective way to generate peptides with various bioactive properties. Furthermore, enzyme immobilization is a way to recycle enzymes for the future uses. The objective of this study was to investigate the effects of neutrase and thermolysin immobilization on the enzyme properties and in vitro anti-diabetic properties of intestinal digests achieved by the simulated digestion of egg white protein hydrolysates.
Material and Methods: Neutrase and thermolysin were immobilized on cellulose-coated magnetite nanoparticles. Then, enzyme activity, thermal resistance, reusability and optimum conditions of the egg white protein hydrolysis were assessed. Egg white protein hydrolysates were then digested in vitro and inhibitory activities of the intestinal digests against dipeptidyl peptidase IV (DPP-IV) and α-glucosidase were investigated.
Results and Conclusion: Enzymes immobilization resulted in increases in the thermal stabilities of them. Optimum temperatures for the egg white protein hydrolysis increased by 4.0 and 3.2 °C for neutrase and thermolysin, respectively. Digests from the hydrolysates of free neutrase effectively inhibited DPP-IV and α-glucosidase by 17.9 and 29.7%, respectively. These values for the hydrolysates released by the free thermolysin were higher (37.2 and 35.1%, respectively). The enzyme immobilization resulted in a 4.4% decrease in DPP-IV inhibitory activities of the digests for the hydrolysates from neutrase and a 28.6% decrease for those from thermolysin. Decreases in α-glucosidase inhibition due to the immobilization included 9.8% for neutrase and 12.2% for thermolysin for the digests from the hydrolysates. Based on the results from the current study, hydrolysates from the egg white proteins achieved by the free and immobilized neutrase and thermolysin can be used in formulations of the functional foods and nutraceuticals with multifunctional properties.
- α-Glucosidase
- Dipeptidyl peptidase IV
- Magnetite nanoparticles
- Neutrase
- Thermolysin
How to Cite
References
Mehta J, Bhardwaj N, Bhardwaj SK, Kim K-H, Deep A. Recent advances in enzyme immobilization techniques: Metal-organic frameworks as novel substrates. Coordin. Chem. Rev. 2016;322:30-40. https://doi.org/10.1016/j.ccr.2016.05.007
Vaghari H, Jafarizadeh-Malmiri H, Mohammadlou M, Berenjian A, Anarjan N, Jafari N, et al. Application of magnetic nanoparticles in smart enzyme immobilization. Biotechnol. Lett. 2016;38(2):223-33. https://doi.org/10.1007/s10529-015-1977-z
Fortes CC, Daniel-da-Silva AL, Xavier AM, Tavares AP. Optimization of enzyme immobilization on functionalized magnetic nanoparticles for laccase biocatalytic reactions. Chem. Eng. Process. 2017;117:1-8. https://doi.org/10.1016/j.cep.2017.03.009
Aslani E, Abri A, Pazhang M. Immobilization of trypsin onto Fe3O4@ SiO2–NH2 and study of its activity and stability. Colloids. Surface. B. 2018;170:553-62. https://doi.org/10.1016/j.colsurfb.2018.06.022
Li R, Fu G, Liu C, McClements DJ, Wan Y, Wang S, et al. Tannase immobilisation by amino-functionalised magnetic Fe3O4-chitosan nanoparticles and its application in tea infusion. Int. J. Bio. Macromol. 2018;114:1134-43. https://doi.org/10.1016/j.ijbiomac.2018.03.077
Chen C, Chi Y-J, Zhao M-Y, Lv L. Purification and identification of antioxidant peptides from egg white protein hydrolysate. Amino. Acids. 2012;43(1):457-66. https://doi.org/10.1007/s00726-011-1102-0
Zambrowicz A, Pokora M, Setner B, Dąbrowska A, Szołtysik M, Babij K, et al. Multifunctional peptides derived from an egg yolk protein hydrolysate: isolation and characterization. Amino. Acids. 2015;47(2):369-80. https://doi.org/10.1007/s00726-014-1869-x
Yu Z, Yin Y, Zhao W, Yu Y, Liu B, Liu J, et al. Novel peptides derived from egg white protein inhibiting alpha-glucosidase. Food. Chem.. 2011;129(4):1376-82. https://doi.org/10.1016/j.foodchem.2011.05.067
Cho D-Y, Jo K, Cho SY, Kim JM, Lim K, Suh HJ, et al. Antioxidant effect and functional properties of hydrolysates derived from egg-white protein. Korean. J. Food. Sci. An. 2014;34(3):362. https://doi.org/10.5851/kosfa.2014.34.3.362
Ortega N, Perez-Mateos M, Pilar MaC, Busto MaD. Neutrase immobilization on alginate− glutaraldehyde beads by covalent attachment. J. Agr. Food. Chem. 2008;57(1):109-15. https://doi.org/10.1021/jf8015738
Chen F, Zhang F, Du F, Wang A, Gao W, Wang Q, et al. A novel and efficient method for the immobilization of thermolysin using sodium chloride salting-in and consecutive microwave irradiation. Bioresource. Technol. 2012;115:158-63. https://doi.org/10.1016/j.biortech.2011.11.059
Rocha-Martin J, Fernández-Lorente G, Guisan JM. Sequential hydrolysis of commercial casein hydrolysate by immobilized trypsin and thermolysin to produce bioactive phosphopeptides. Biocatal. Biotransfor. 2018;36(2):159-71. https://doi.org/10.1080/10242422.2017.1308499
Namdeo M, Bajpai S. Immobilization of α-amylase onto cellulose-coated magnetite (CCM) nanoparticles and preliminary starch degradation study. J. Mol. Catal. B-Enzym. 2009;59(1-3):134-9. https://doi.org/10.1016/j.molcatb.2009.02.005
Jain TK, Morales MA, Sahoo SK, Leslie-Pelecky DL, Labhasetwar V. Iron oxide nanoparticles for sustained delivery of anticancer agents. Mol. Pharm. 2005;2(3):194-205. https://doi.org/10.1021/mp0500014
Yang A, Long C, Xia J, Tong P, Cheng Y, Wang Y, et al. Enzymatic characterisation of the immobilised Alcalase to hydrolyse egg white protein for potential allergenicity reduction. J. Sci. Food. Agr. 2017;97(1):199-206. https://doi.org/10.1002/jsfa.7712
Lin S, Guo Y, Liu J, You Q, Yin Y, Cheng S. Optimized enzymatic hydrolysis and pulsed electric field treatment for production of antioxidant peptides from egg white protein. Afr. J. Biotechnol. 2011;10(55):11648-57.
del Mar Contreras M, Hernández-Ledesma B, Amigo L, Martín-Álvarez PJ, Recio I. Production of antioxidant hydrolyzates from a whey protein concentrate with thermolysin: Optimization by response surface methodology. LWT-Food. Sci. Technol. 2011;44(1):9-15. https://doi.org/10.1016/j.lwt.2010.06.017
Nielsen P, Petersen D, Dambmann C. Improved method for determining food protein degree of hydrolysis. J. Food. Sci. 2001;66(5):642-6. https://doi.org/10.1111/j.1365-2621.2001.tb04614.x
Minekus M, Alminger M, Alvito P, Ballance S, Bohn T, Bourlieu C, et al. A standardised static in vitro digestion method suitable for food–an international consensus. Food. Funct. 2014;5(6):1113-24. https://doi.org/10.1039/C3FO60702J
Kehinde BA, Sharma P. Recently isolated antidiabetic hydrolysates and peptides from multiple food sources: a review. Crit. Rev. Food. Sci. Nutr. 2020;60(2):322-40. https://doi.org/10.1080/10408398.2018.1528206
Lacroix IM, Li-Chan EC. Dipeptidyl peptidase-IV inhibitory activity of dairy protein hydrolysates. Int. Dairy. J. 2012;25(2):97-102. https://doi.org/10.1016/j.idairyj.2012.01.003
Mojica L, de Mejía EG. Optimization of enzymatic production of anti-diabetic peptides from black bean (Phaseolus vulgaris L.) proteins, their characterization and biological potential. Food. Funct. 2016;7(2):713-27. https://doi.org/10.1039/C5FO01204J
Jia N, Li S-M, Ma M-G, Zhu J-F, Sun R-C. Synthesis and characterization of cellulose-silica composite fiber in ethanol/water mixed solvents. BioResources. 2011;6(2):1186-95.
Kassanov B, Wang J, Fu Y, Chang J. Cellulose enzymatic saccharification and preparation of 5-hydroxymethylfurfural based on bamboo hydrolysis residue separation in ionic liquids. RSC. Adv. 2017;7(49):30755-62. https://doi.org/10.1039/C7RA05020H
Jensen JK, Rubio JM, Engelsen SB, van den Berg F. Protein residual fouling identification on UF membranes using ATR-FT-IR and multivariate curve resolution. Chemometr. Intell. Lab. 2015;144:39-47. https://doi.org/10.1016/j.chemolab.2015.03.006
Xin Bj, Si Sf, Xing Gw. Protease Immobilization on γ‐Fe2O3/Fe3O4 Magnetic Nanoparticles for the Synthesis of Oligopeptides in Organic Solvents. Chem. Asian. J. 2010;5(6):1389-94. https://doi.org/10.1002/asia.200900696
Dal Magro L, Kornecki JF, Klein MP, Rodrigues RC, Fernandez-Lafuente R. Optimized immobilization of polygalacturonase from Aspergillus niger following different protocols: Improved stability and activity under drastic conditions. Int. J. Biol. Macromol. 2019;138:234-43. https://doi.org/10.1016/j.ijbiomac.2019.07.092
Rao RS, Borkar PS, Khobragade C, Sagar A. Enzymatic activities of proteases immobilized on tri (4-formyl phenoxy) cyanurate. Enzyme. Microb. Tech. 2006;39(4):958-62. https://doi.org/10.1016/j.enzmictec.2006.03.020
Sheldon RA. Enzyme immobilization: the quest for optimum performance. Adv. Synth. Catal. 2007;349(8‐9):1289-307. https://doi.org/10.1002/adsc.200700082
Saqib AA, Siddiqui KS. How to calculate thermostability of enzymes using a simple approach. Biochem. Mol. Biol. Educ. 2018;46(4):398-402. https://doi.org/10.1002/bmb.21127
Zhang Y, Tang Z, Wang J, Wu H, Lin C-T, Lin Y. Apoferritin nanoparticle: a novel and biocompatible carrier for enzyme immobilization with enhanced activity and stability. J. Mater. Chem. 2011;21(43):17468-75. https://doi.org/10.1039/C1JM11598G
Mansfeld J, Vriend G, Van den Burg B, Eijsink VG, Ulbrich-Hofmann R. Probing the unfolding region in a thermolysin-like protease by site-specific immobilization. Biochemistry. 1999;38(26):8240-5. https://doi.org/10.1021/bi990008p
Wu X-c, Zhang Y, Wu C-y, Wu H-x. Preparation and characterization of magnetic Fe3O4/CRGO nanocomposites for enzyme immobilization. T. Nonferr. Metal. Soc. 2012;22:s162-s8. https://doi.org/10.1016/S1003-6326(12)61703-8
Pal A, Khanum F. Covalent immobilization of xylanase on glutaraldehyde activated alginate beads using response surface methodology: characterization of immobilized enzyme. Process. Biochem. 2011;46(6):1315-22. https://doi.org/10.1016/j.procbio.2011.02.024
Power O, Nongonierma AB, Jakeman P, Fitzgerald RJ. Food protein hydrolysates as a source of dipeptidyl peptidase IV inhibitory peptides for the management of type 2 diabetes. Proc. Nutr. Soc. 2014;73(1):34-46. https://doi.org/10.1017/S0029665113003601
Yu Z, Yin Y, Zhao W, Liu J, Chen F. Anti-diabetic activity peptides from albumin against α-glucosidase and α-amylase. Food. Chem. 2012;135(3):2078-85. https://doi.org/10.1016/j.foodchem.2012.06.088
- Abstract Viewed: 569 times
- PDF Downloaded: 532 times
