Optimizing Submerged Cultivation for the Production of Red Pigments by Monascus purpureus on Soybean Meals using Response Surface Methodology
Applied Food Biotechnology,
Vol. 7 No. 3 (2020),
10 Khordad 2020
,
Page 143-152
https://doi.org/10.22037/afb.v7i3.28931
Abstract
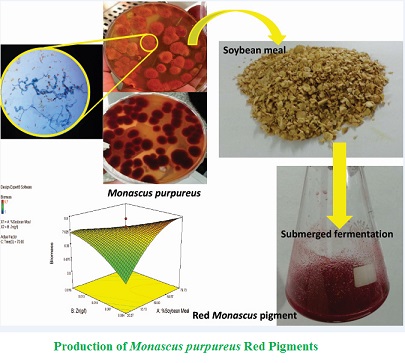
Background and objective: Monascus purpureus is a filamentous fungus with ability to produce pigments with therapeutic properties. Red pigments are especially used as additives, antioxidants, preservatives and substitutions for nitrites in food technology. To decrease fermentation costs, agro-industrial wastes such as soybean meals have been used as substrates. In the current study, red pigment production by Monascus purpureus on soybean meals was optimized.
Material and methods: In this study, red Monascus pigment production by Monascus purpureus ATCC 16362 was carried out under submerged fermentation using soybean meals as nitrogen sources to replace yeast extracts. Central composite design was used to assess the optimum level of soybean meal replacement (0-100%), ZnSO4.7H2O concentration (0-0.02
g l-1) and thermal stress time of spore suspension at 70°C (50-90 s). Red Monascus pigment and biomass productions were assessed as dependent responses.
Results and conclusion: The maximum production of red Monascus pigment (4.54 AU ml-1) was achieved under conditions of soybean meal replacement of 79.72%, ZnSO4.7H2O concentration of 0-0.02 g l-1 and thermal stress time of spore suspension of 81.89 s. The average yield of red Monascus pigment, conversion factor of biomass in red pigment YP/X and cell productivity included 0.324 AU ml-1 day-1, 1.10 AU L g-1 and 0.292 g l-1 day-1, respectively. Results of the current study have demonstrated that combination of soybean meal and yeast extract as nitrogen source is beneficial for the production of red Monascus pigment by Monascus purpureus.
Conflict of interest: The authors declare no conflict of interest.
- ▪ Central composite design ▪ Monascus purpureus ▪ Red pigment ▪ Response surface method ▪ Soybean meal
How to Cite
References
Panesar R, Kaur S, Panesar PS. Production of microbial pigments utilizing agro-industrial waste: A review. J Curr Opin Food Sci. 2015; 1: 70–76.
doi: 10.1016/j.cofs.2014.12.002
Azlina W, Arul C, Dufoss L. Current perspective of yellowish-orange pigments from microorganisms: A review. J Clean. Prod. 2018; 180:168–182.
doi: 10.1016/j.jclepro.2018.01.093
Agboyibor C, Kong W, Chen D, Zhang A, Niu, S. Monascus pigments production, composition ,bioactivity and its application: A review. J ISBAB. 2018 ;16: 433–447.
doi: 10.1016/j.bcab.2018.09.012
Singgih M, Saraswaty V, Ratnaningrum D, Priatni S, Damayanti S. The influence of temperature and ethanol concentration in monacolin K extraction from Monascus fermented rice. J LWT. 2014; 9: 242–247.
doi: 10.1016/j.proche.2014.05.029
Farhadi G B N, Khosravi-Darani K, Naser nejad B. Enhancement of xanthan production on date extract using response surface methodology. Chem. Asian J. 2012; 24: 3887-3890.
Zhu Z, Zhang F,Wei Z, Ran W, Shen Q. The usage of rice straw as a major substrate for the production of surfactin by Bacillus amyloliquefaciens XZ-173 in solid-state fermentation. J. Environ. Manage. 2013; 127: 96-102.
doi: 10.1016/j.jenvman.2013.04.017
Seyede, Reihani F S, Khosravi-Darani K. Mycoprotein production from date waste using Fusarium venenatum in a submerged culture. Appl. Food Biotechnol. 2018; 5: 243-252.
doi: 10.22037/afb.v5i4.23139
Hashempour-Baltork F, Khosravi-Darani K, Hosseini H, Farshi P, S. Reihani S.F. Mycoprotein as safe meat substitutes: A review. J. Clean. Prod. 2020; 253: 119958.
doi: https://doi.org/10.1016/j.jclepro.2020.119958
Mojumdar A, Deka J. Recycling agro industrial waste to produce amylase and characterizing amylase–gold nanoparticle composite. Int J Recycle Org Waste Agri. 2019 ;136: 1–7.
doi: 10.1007/s40093-019-00298-4
Sharma R, Ghoshal G. Optimization of carotenoids production by Rhodotorula mucilaginosa (MTCC-1403) using agro-industrial waste in bioreactor: A statistical approach. J Biotechnol Reports. 2020 ;25: e00407.
doi: 10.1016/j.btre.2019.e00407
Bakhshi F, Jahadi M. Optimization of orange pigment production by Monascus purpureus from date palms by response surface methodology. J Food Microbiol. 2018; 4: 31-39. (Persian)
Silveira ST, Daroit DJ, Brandeli A. Pigment production by Monascus purpureus in grape waste using factorial design. J LWT. 2008; 41: 170–174.
doi: 10.1016/j.lwt.2007.01.013
Haque MA, Kachrimanidou V, Koutinas A. Valorization of bakery waste for biocolorant and enzyme production by Monascus purpureus. J Biotechnol. 2016;231: 55–64.
doi: 10.1016/j.jbiotec.2016.05.003
Kantifedaki A, Kachrimanidou V,Mallouchos A, Papanikolaou S, Koutinas A.A. Orange processing waste valorisation for the production of bio-based pigments using the fungal strains Monascus purpureus and Penicillium purpurogenum. J Clean. Prod. 2018;185: 882–890.
doi: 10.1016/j.jclepro.2018.03.032
Chiu S.W, Chan S.M. Production of pigments by Monascus purpureus using sugar-cane bagasse in roller bottle cultures. JMB. 1992;8: 68-70.
doi: 10.1007/BF01200689
Silveira S T, Daroit D J, Sant’Anna V,Brandelli A. Stability modeling of red pigments produced by Monascus purpureus in submerged cultivations with sugarcane bagasse. J Food Bioproc Tech. 2013;6: 1007-1114.
doi: 10.1007/s11947-011-0710-8
Duraklı-VelioğluS,Boyacı İ.H, Şimşek O,Gümüş T. Optimizing a submerged Monascus cultivation for production of red pigment with bug damaged wheat using artificial neural networks. J Food Sci Biotechnol. 2013;22: 1639-1648.
doi: 10.1007/s10068-013-0261-z
Yuliana A, Singgih M, Julianti E, Blanc P. Derivates of azaphilone Monascus pigments: A review. J Biocatal agric biotechnol. 2013; 40: 169–181.
doi: http://dx.doi.org/10.1016/j.bcab.2016.12.014
Chen M, Johns MR. 1993. Effect of pH and nitrogen source on pigment production by Monascus purpureus. J Appl Microbiol Biotechnol. 1993; 40: 132 – 138.
doi: https://doi.org/10.1007/BF00170441
Bau YS, Wong HC. Zinc effects on growth, pigmentation and antibacterial activity of Monascus purpureus. J Physiol. Plant.1979; 46: 63-67.
doi: 10.1111/j.1399-3054. 1979.tb03187.x
da Costa J P V, Vendruscolo F. Production of red pigments by Monascus rubber CCT 3802 using lactose as substrate. J Agric. Biotech. 2017;11: 50–55.
doi: 10.1016/j.bcab.2017.05.010
Meinicke R M, Vendruscolo F, Moritz D E, de Oliveira D, Schmidell W, Samohyl R W and Ninow J L. Potential use of glycerol as substrate for the production of red pigments by Munascus rubber in submerged fermentation. Int J Innov Res Sci Eng Technol. 2012; 1:238-242.
doi: 10.1016/j.bcab.2012.03.001
Seyedin A, Yazdian F, Hatamian-Zarmi A, Rasekhi B, Mir-derikvand M. Natural red pigment production by Monascus purpureus: Bioreactor yield improvement through statistical analysis. J Appl. Food Biotechnol. 2015; 2(2): 23-30.
doi: 10.22037/afb.v.2i2.7457
Sharmila G, Nidhi B, Muthukumaran C. Sequential statistical optimization of red pigment production by Monascus purpureus (MTCC 369) using potato powder. J Ind Crops and Prod. 2013;44: 158–164.
doi: 10.1016/j.indcrop.2012.11.007
Babitha S, Soccol C. R, Pandey A. Effect of stress on growth , pigment production and morphology of Monascus sp . in solid cultures. J Basic Microbiol. 2007;47: 118–126.
doi: 10.1002/jobm.200610261
Thakur, Nemade S.N,Sharanappa A . Solid state fermentation of overheated soybean meal (waste) for production of protease using Aspergillus Oryzae. Int J Innov Res Sci Eng Technol. 2015; 4:18456-18461.
doi: 10.15680/IJIRSET.2015.0401008
Silva F, Daniel F. Characterization and stability of bioactive compounds from soybean meal. J Food Sci Technol. 2015; 63: 992-1000.
doi: 10.1016/j.lwt.2015.04.032
Zhang K, Yu C, Yang S. Effects of soybean meal hydrolysate as the nitrogen source on seed culture morphology and fumaric acid production by Rhizopus oryzae. J Process Biochem.2015;50(2): 173–179.
doi: 10.1016/j.procbio.2014.12.015
Lujan-Rhenals D, Morawicki R, Gbur E, Ricke S. Fermentation of soybean meal hydrolyzates with Saccharomyces cerevisiae and Zymomonasmobilis for ethanol production. J Food Sci. 2015;80: 1512–1518.
doi: 10.1111/1750-3841.12907
Zhang J, Wang Y,LuL, Zhang B, Xu G. Enhanced production of Monacolin K by addition of precursors and surfactants in submerged fermentation of Monascus purpureus 9901. J Biotecnol App Bioc. 2014;61(2): 202-207.
doi: 10.1002/bab.1154
Kawahara Y, Kikuchi Y, Kurahashi, O, Kimura E, Nakamatsu T, Goto S. Stress-resistant microorganism and method of producing fermentative product. United
States Patent. 2002; 6: 338-956.
Stroher R, Stenzel M, Pereira N C, Zanin G M. Enzymatic extraction of protein from toasted and not toasted soybean meal. J Food Sci. 2011; 1: 463-469.
doi: 10.1016/j.profoo.2011.09.071
Kongruang S. Growth kinetic of biopigment production by Thai isolated Monascus purpureus in a stirred tank bioreactor. J Micro Biotechnol. 2018;38: 93–99.
doi: 10.1007/s10295-010-0834-2
Jahadi, M. Khosravi-Darani, K., Ehsani, MR. Mozafari, M.R. Saboury, A. S. Pourhosseini, P. S. The encapsulation of flavourzyme in nanoliposome by heating method. J Food Sc and Technol. 2015;52( 4): 2063-2072.
doi: 10.1007/s13197-013-1243-0
Hajjaj H, Franc¸ois J-M,GomaG,BlancP J. Effect of amino acids on red pigments and citrinin production in Monascus rubber. J Food Sci. 2012;77: 156–159.
doi: 10.1111/j.1750-3841.2011.02579.x
Adiga, P. R., Sivarama Sastry, K., VenkatasuBbramanyan, V. &Sarma, P. S. Interrelationships in trace-element metabolism in Aspergillus niger. J Biochem. 1961;81Z:545-550.
doi: 10.1042/bj0810545
Vendruscolo F, Willibaldo S, Oliveira D, Ninow J. Kinetic of orange pigment production from Monascus ruber on submerged fermentation. J Bioproc Biosyt Eng. 2017; 40(1): 115-121.
doi: 10.1007/s00449-016-1679-5
Abrashev R, Dolashka P, Cristova R, Stefanova L,Angelova M. Role of antioxidant enzymes in survival of conidiospores of Aspergillus niger 26 under conditions of temperature stress. J Appl. Microbiol. 2005;99(4): 902–909.
doi: 10.1111/j.1365-2672.2005.02669.x
Fridovich I. Superoxide radical and superoxide dismutases. J Annu. Rev. Biochem. 1995; 64:97–112.
Li Q, Harvey LM, McNeil B. Oxidative stress in industrial fungi. J Crit Rev Biotechnol. 2009; 29:199-213.
doi: 10.1080/07388550903004795
Edward G, Perkins. Composition of soybeans and soybean products. In: David R, Erickson. Practical Handbook of Soybean Processing and Utilization, 1st Edition. Academic Press and AOCS Press, 1995:9-28.
doi:10.1016/B978-0-935315-63-9.50006-1
Gadd, G.M. ‘‘Interactions of fungi with toxic metals’’. J New Phytol. 1993;124:25–60.
doi: 10.1007/978-1-4899-0981-7_28
Broder C.U, Koehler P.E. Pigments produced by Monascus purpureus with regard to quality and quantity. J Food Sci. 1980;45: 567-569.
- Abstract Viewed: 1596 times
- PDF Downloaded: 703 times
