Effects of Major Ingredients in Cattle Milk on Enzyme Kinetics of Recombinant β-galactosidase (BGalP) Expressed in Pichia pastoris
Applied Food Biotechnology,
Vol. 5 No. 4 (2018),
17 September 2018
,
Page 205-212
https://doi.org/10.22037/afb.v5i4.22107
Abstract
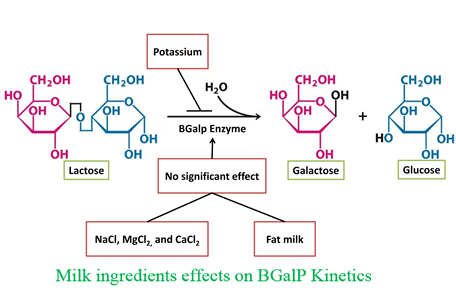
Background and objective: β-galactosidase enzymes hydrolyze lactose into glucose and galactose for production of lactose free dairy products. However, different ions and fat content in milk may act as the inhibitor or activator for β-galactosidase enzymes. A cold-active β-galactosidase enzyme (BGalP), originally from Planococcus sp. L4, was previously expressed in Pichia pastoris to perform lactose hydrolysis in the refrigerated milk. In this study, the effects of milks major ingredients were evaluated on the enzymatic kinetics to confirm its capacity for hydrolyzing milk lactose.
Material and methods: The activity was determined in different concentrations of NaCl, KCl, MgCl2, and CaCl2 as well as in the milk with low, medium or high-fat content. In these experiments ortho-Nitrophenyl β-galactoside was used as the substrate. Additionally, glucose was measured as the product after incubation of milk with BGalP enzyme for 24 h at room temperature.
Results and conclusion: This study demonstrated that ions and fat content did not adversely affect the enzyme activity in the concentration corresponding to the milk contents. Ca (27.5-32.5 mM), Cl (25.3-30.9 mM), Na (15.2-39.1 mM) and Mg (3.75-5.83 mM) had no inhibitory effects, but KCl decreased the enzyme activity. Since Cl existed in MgCl2, and CaCl2 exerted no inhibitory effects, it can be concluded that inhibitory effects of KCl resulted from potassium rather than chloride. The results indicate that BGalP enzyme was not inhibited by milks major ingredients and has the potential to be used for the production of lactose-free dairy products.
Conflict of interest: The authors declare no conflict of interest.
- ▪ BGalP ▪ Enzyme food biotechnology ▪ Kinetics ▪ Milk
How to Cite
References
Haug A, Hostmark A, Harstad OM. Bovine milk in human nutrition–a review. Lipids Health Dis. 2007;6(25):1-16.DOI: https://doi.org/10.1186/1476-511X-6-25
Baadkar SV, Mukherjee MS, Lele SS. Study on influence of age, gender and genetic variants on lactose intolerance and its impact on milk intake in adult Asian Indians. Ann Hum Biol. 2014;41(6):548-53.DOI: 10.3109/03014460.2014.902992
Estevez CA, Isasi JR, Larraneta E, Velaz I. Release of beta-galactosidase from poloxamine/alpha-cyclodextrin hydrogels. Beilstein J Org Chem. 2014;10:3127-35. DOI: 10.3762/bjoc.10.330
Rosado JL. [Lactose intolerance]. Gac Med Mex. 2016;152 Suppl 1:67-73
Abbasalizadeh S, Hejazi MA, Hajiabbas MP. Kinetics of β-galactosidase Production by Lactobacillus bulgaricus During pH Controlled Batch Fermentation in Three Commercial Bulk Starter Media. Appl Food Biotechnol. 2015;2(4):39-47. DOI: http://dx.doi.org/10.22037/afb.v2i4.9512
Husain Q. β Galactosidases and their potential applications: a review. Crit Rev Biotechnol. 2010;30(1):41-62.DOI: https://doi.org/10.3109/07388550903330497
Zhou QZK, Chen XD. Effects of temperature and pH on the catalytic activity of the immobilized β-galactosidase from Kluyveromyces lactis. Biochem Eng J. 2001;9(1):33-40.DOI: http://dx.doi.org/10.1016/S1369-703X(01)00118-8
Katrolia P, Yan Q, Jia H, Li Y, Jiang Z, Song C. Molecular cloning and high-level expression of a β-galactosidase gene from Paecilomyces aerugineus in Pichia pastoris. J Mol Catal B Enzym 2011;69(3):112-9.DOI: https://doi.org/10.1016/j.molcatb.2011.01.004
Kim CS, Ji ES, Oh DK. A new kinetic model of recombinant beta-galactosidase from Kluyveromyces lactis for both hydrolysis and transgalactosylation reactions. Biochem Biophys Res Commun. 2004;316(3):738-43.DOI: 10.1016/j.bbrc.2004.02.118
Kim CS, Ji ES, Oh DK. Expression and characterization of Kluyveromyces lactis beta-galactosidase in Escherichia coli. Biotechnol Lett. 2003;25(20):1769-74.DOI: https://doi.org/10.1023/A:1026092029785
Hu JM, Li H, Cao LX, Wu PC, Zhang CT, Sang SL, et al. Molecular cloning and characterization of the gene encoding cold-active β-galactosidase from a psychrotrophic and halotolerant Planococcus sp. L4. J Agric Food Chem. 2007;55(6):2217-24.DOI: 10.1021/jf062910r
Juajun O, Nguyen TH, Maischberger T, Iqbal S, Haltrich D, Yamabhai M. Cloning, purification, and characterization of beta-galactosidase from Bacillus licheniformis DSM 13. Appl Microbiol Biotechnol. 2011;89(3):645-54.DOI: 10.1007/s00253-010-2862-2
Brandao RL, Nicoli JR, Figueiredo AF. Purification and characterization of a beta-galactosidase from Fusarium oxysporum var. lini. J Dairy Sci. 1987;70(7):1331-7
Maischberger T, Leitner E, Nitisinprasert S, Juajun O, Yamabhai M, Nguyen TH, et al. Beta-galactosidase from Lactobacillus pentosus: purification, characterization and formation of galacto-oligosaccharides. Biotechnol J. 2010;5(8):838-47.DOI: 10.1002/biot.201000126
Vella M, Greenwell P. Purification and partial characterization of beta-galactosidase from Tritrichomonas foetus. Glycoconj J. 1997;14(7):883-7
Lomer MC, Parkes GC, Sanderson JD. Review article: lactose intolerance in clinical practice--myths and realities. Aliment Pharmacol Ther. 2008;27(2):93-103.DOI: 10.1111/j.1365-2036.2007.03557.x
Hildebrandt P, Wanarska M, Kur J. A new cold-adapted beta-D-galactosidase from the Antarctic Arthrobacter sp. 32c - gene cloning, overexpression, purification and properties. BMC Microbiol. 2009;9:151.DOI: 10.1186/1471-2180-9-151
Mahdian SM, Karimi E, Tanipour MH, Parizadeh SM, Ghayour-Mobarhan M, Bazaz MM, et al. Expression of a functional cold active beta-galactosidase from Planococcus sp-L4 in Pichia pastoris. Protein Expr Purif. 2016;125:19-25.DOI: 10.1016/j.pep.2015.09.008
Juers DH, Matthews BW, Huber RE. LacZ β‐galactosidase: structure and function of an enzyme of historical and molecular biological importance. Protein Sci. 2012;21(12):1792-807.DOI: https://doi.org/10.1002/pro.2165
Van Laere KM, Abee T, Schols HA, Beldman G, Voragen AG. Characterization of a Novel β-Galactosidase fromBifidobacterium adolescentis DSM 20083 Active towards Transgalactooligosaccharides. Appl Environ Microbiol. 2000;66(4):1379-84.DOI: 10.1128/AEM.66.4.1379-1384.2000
Reithel F, Kim JC. Studies on the β-galactosidase isolated from Escherichia coli ML 308. I. The effect of some ions on enzymic activity. Arch Biochem Biophys. 1960;90(2):271-7
- Abstract Viewed: 890 times
- PDF Downloaded: 485 times