Partial Proteomics Analysis of Montivipera raddei Venom Partial proteomics of Montivipera raddei venom
Trends in Peptide and Protein Sciences,
Vol. 8 No. 1 (2023),
24 December 2023
,
Page 1-5 (e2)
https://doi.org/10.22037/tpps.v8i1.41610
Abstract
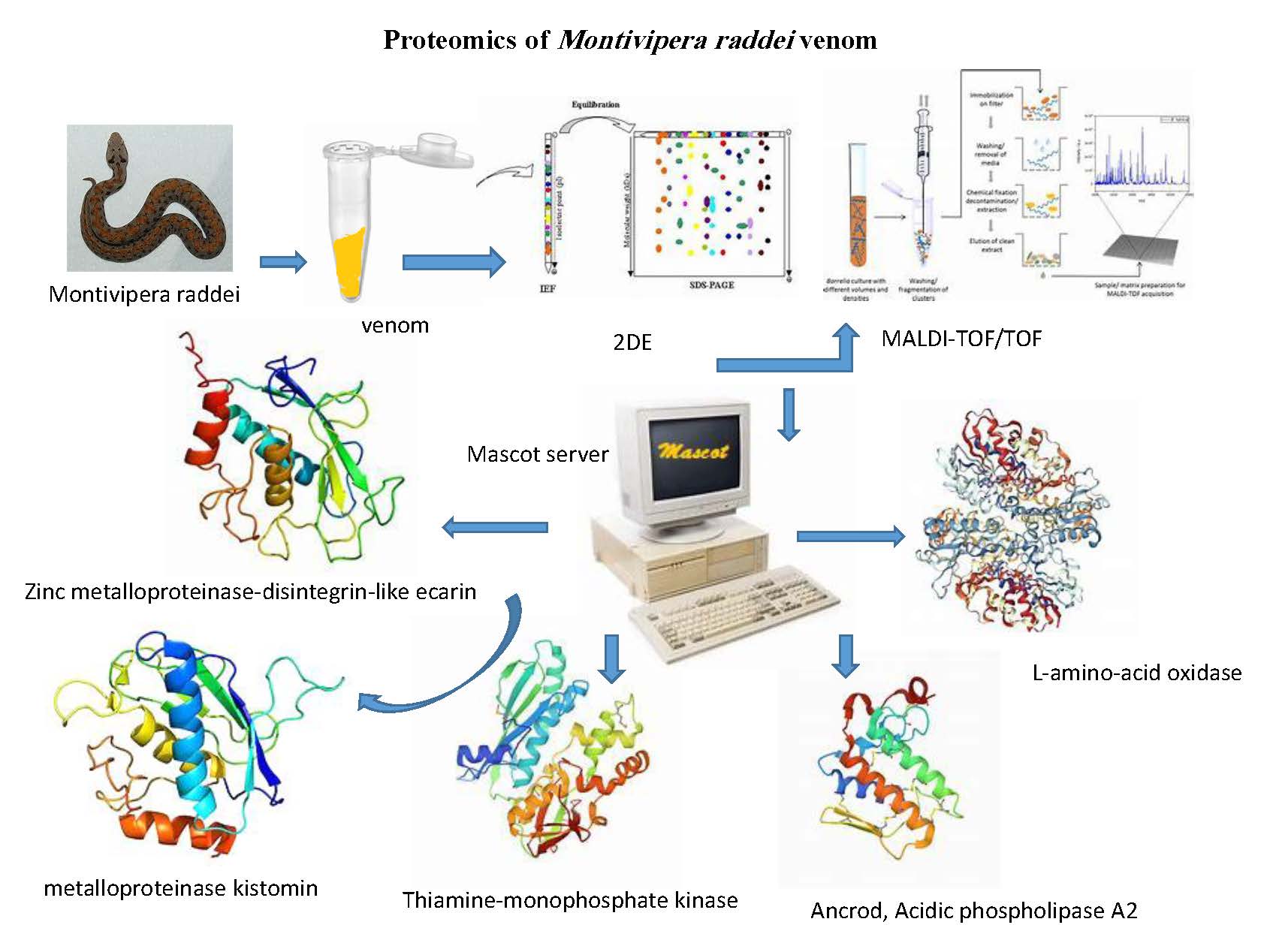
The snake venom is a potent source of a variety of drugs and therapeutic components. This study aimed to isolate and characterize some of proteins in Montivipera raddei venom. The protein bands and spots obtained by SDS-PAGE and two-dimensional electrophoresis were analyzed. The separated protein spots based on isoelectric point and molecular weight were scattered in the 15 to 66 kDa ranges and pI from 5 to 8. Six proteins was more extensively characterized by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF/TOF) analysis. These characterized proteins included Zinc metalloproteinase-disintegrin-like ecarin, L-amino-acid oxidase, metalloproteinase kistomin, Thiamine-monophosphate kinase, Ancrod, Acidic phospholipase A2.
HIGHLIGHTS
- Identification of several proteins from Montivipera raddei venom.
- Characterization of the six high concentrated proteins by MALDI-TOF/TOF spectroscopy in venom.
- Zinc metalloproteinase-disintegrin-like ecarin, L-amino-acid oxidase, metalloproteinase kistomin, Thiamine-monophosphate kinase, Ancrod, Acidic phospholipase A2 were identified in venom of Montivipera raddei.
- 2D-electrophoresis
- Mass spectroscopy
- Montivipera raddei
- Proteomics
- Venom
How to Cite
References
Bradford, M.M. (1976). ″A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding.″ Analytical Biochemistry. 72:248-254. DOI: https://doi.org/10.1016/0003-2697(76)90527-3.
Bunai, K. and K. Yamane, (2005). ″Effectiveness and limitation of two-dimensional gel electrophoresis in bacterial membrane protein proteomics and perspectives.″ Journal of Chromatography B: Analytical Technologies in the Biomedical and Life Sciences, 815(1-2): 227-236. DOI: https://doi.org/10.1016/j.jchromb.2004.08.030.
Casais-e-Silva, L. L., Teixeira, C. F., Lebrun, I., Lomonte, B., Alape-Giron, A. and J. M. Gutiérrez, (2016). ″Lemnitoxin, the major component of Micrurus lemniscatus coral snake venom, is a myotoxic and pro-inflammatory phospholipase A2.″ Toxicology Letters, 257: 60-71. DOI: https://doi.org/10.1016/j.toxlet.2016.06.005.
Chen, L., Xia, H., Wang, Y., Chen, K., Qin, L. and B. Wang, (2013). ″Proteomic profiling of liver from Elaphe taeniura, a common snake in eastern and southeastern Asia.″ Genetic Molecular Biology, 36(3): 438-447. DOI: https://doi.org/10.1590/S1415-47572013000300020.
Costa, S., Camargo, E. and E. Antunes, (2017). ″Inflammatory action of secretory phospholipases A2 from snake venoms.″ In: Toxins and Drug Discovery; Gopalakrishnakone, P., Cruz, L., Luo, S., (Eds), Springer, Dordrecht. pp. 35-52. DOI: https://doi.org/10.1007/978-94-007-6452-1_10.
Dempfle, C.-E., Alesci, S., Kucher, K., Müller-Peltzer, H., Rübsamen, K. and M. Borggrefe, (2001). ″Plasminogen activation without changes in tPA and PAI-1 in response to subcutaneous administration of ancrod.″ Thrombosis Research Journal, 104(6): 433-438. DOI: https://doi.org/10.1016/S0049-3848(01)00391-7.
Fox, J.W. and S.M. Serrano, (2008). ″Exploring snake venom proteomes: multifaceted analyses for complex toxin mixtures.″ Proteomics, 8(4): 909-920. DOI: https://doi.org/10.1002/pmic.200700777.
Harris, J.B. and T. Scott-Davey, (2013). ″Secreted phospholipases A2 of snake venoms: effects on the peripheral neuromuscular system with comments on the role of phospholipases A2 in disorders of the CNS and their uses in industry.″ Toxins, 5(12): 2533-2571. DOI: https://doi.org/10.3390/toxins5122533.
Kini, R.M. (2005). ″Serine proteases affecting blood coagulation and fibrinolysis from snake venoms.″ Pathophysiology of Haemostasis and Thrombosis, 34(4-5):200-204. DOI: https://doi.org/10.1159/000092424.
Kini, R.M. and C.Y. Koh, (2016). ″Metalloproteases affecting blood coagulation, fibrinolysis and platelet aggregation from snake venoms: definition and nomenclature of interaction sites.″ Toxins, 8(10): 284. DOI: https://doi.org/10.3390/toxins8100284.
Mackessy, S.P., Sixberry, N.M., Heyborne, W.H. and T. Fritts, (2006). ″Venom of the Brown Treesnake, Boiga irregularis: ontogenetic shifts and taxa-specific toxicity.″ Toxicon, 47(5): 537-548. DOI: https://doi.org/10.1016/j.toxicon.2006.01.007.
Munawar, A., Ali, S. A., Akrem, A., and C. Betzel, (2018). ″Snake venom peptides: Tools of biodiscovery.″ Toxins, 10(11): 474. DOI: https://doi.org/10.3390/toxins10110474.
Moore, G.W. (2007). ″Combining Taipan snake venom time/Ecarin time screening with the mixing studies of conventional assays increases detection rates of lupus anticoagulants in orally anticoagulated patients.″ Thrombosis Journal, 5(1):12. DOI: https://doi.org/10.1186/1477-9560-5-12.
Nishida, S., Fujita, T., Kohno, N., Atoda, H., Morita, T., Takeya, H. and S. Iwanaga, (1995). ″cDNA cloning and deduced amino acid sequence of prothrombin activator (ecarin) from Kenyan Echis carinatus venom.″ Biochemistry, 34(5): 1771-1778. DOI: https://doi.org/10.1021/bi00005a034.
Qin, J., Xu, Z., Shi, D., Chen, D., Dai, J., Teng, H. and Q. Jiang, (2013). ″Deep vein thrombosis after total hip arthroplasty and total knee arthroplasty in patients with previous ischemic stroke.″ Internatinal Journal of Lower Extremity Wounds, 12(4): 316-319. DOI: https://doi.org/10.1177/1534734613493291.
Rao, V. S., Joseph, J. S. and R.M. Kini, (2003). ″Group D prothrombin activators from snake venom are structural homologues of mammalian blood coagulation factor Xa.″ Biochemical Journal, 369(3): 635-642. DOI: https://doi.org/10.1042/bj20020889.
Rosing, J. and G. Tans, (1992). ″Structural and functional properties of snake venom prothrombin activators.″ Toxicon, 30(12): 1515-1527. DOI: https://doi.org/10.1016/0041-0101(92)90023-X.
Sakurai, Y., Shima, M., Matsumoto, T., Takatsuka, H., Nishiya, K., Kasuda, S. and A. Yoshioka, (2003). ″Anticoagulant activity of M-LAO, L-amino acid oxidase purified from Agkistrodon halys blomhoffii, through selective inhibition of factor IX.″ Biochimica et Biophysica Acta - Proteins and Proteomics, 1649(1): 51-57. DOI: https://doi.org/10.1016/S1570-9639(03)00157-2.
Serrano, S. M., Shannon, J. D., Wang, D., Camargo, A. C., and J. W. Fox, (2005). ″A multifaceted analysis of viperid snake venoms by two‐dimensional gel electrophoresis: An approach to understanding venom proteomics.″ Proteomics, 5(2):501-510. DOI: https://doi.org/10.1002/pmic.200400931.
Settembre, E., Begley, T.P. and S.E. Ealick, (2003). ″Structural biology of enzymes of the thiamin biosynthesis pathway.″ Current Opinion in Structural Biology, 13(6): 739-747. DOI: https://doi.org/10.1016/j.sbi.2003.10.006.
Slagboom, J., Mladić, M., Xie, C., Kazandjian, T.D., Vonk, F. and G.W. Somsen, (2020) ″High throughput screening and identification of coagulopathic snake venom proteins and peptides using anofractionation and proteomics approaches.″ PLoS Neglected Tropical Diseases, 14(4): e0007802. DOI: https://doi.org/10.1371/journal.pntd.0007802.
Schieck, A., Kornalik, F. and E. Habermann, (1972). ″The prothrombin-activating principle from Echis carinatus venom.″ Naunyn-Schmiedeberg's Archives of Pharmacology, 272(4): 402-416. DOI: https://doi.org/10.1007/BF00501247.
Tasoulis, T. and G.K. Isbister, (2017). ″A review and database of snake venom proteomes.″ Toxins, 9(9), 290. DOI: https://doi.org/10.3390/toxins9090290.
Vejayan, J., Shin Yee, L., Ponnudurai, G., Ambu, S. and I. Ibrahim, (2010). ″Protein profile analysis of Malaysian snake venoms by two-dimensional gel electrophoresis.″ Journal of Venomous Animals and Toxins Including Tropical Diseases, 16(4):623-630. DOI: https://doi.org/10.1590/S1678-91992010000400013.
Vejayan, J., Khoon, T. L. and H. Ibrahim, (2014). ″Comparative analysis of the venom proteome of four important Malaysian snake species.″ Journal of Venomous Animals and Toxins Including Tropical Diseases, 20(1): 6. DOI: https://doi.org/10.1186/1678-9199-20-6.
- Abstract Viewed: 127 times
- PDF Downloaded: 98 times