Optimization of Antioxidant Activities and Intracellular Polysaccharide Contents Using Agaricus bisporus Extract as Elicitor in Submerged Fermenting Ganoderma lucidum Optimization of IPS production from ganoderma lucidum
Applied Food Biotechnology,
Vol. 8 No. 4 (2021),
2 Mehr 2021
,
Page 297-306
https://doi.org/10.22037/afb.v8i4.35155
Abstract
Background and Objective:
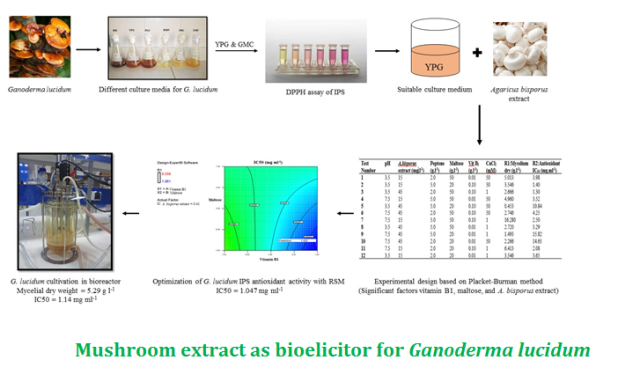
Ganoderma lucidum is one of the medicinal fungi frequently used as supplement. The intracellular polysaccharides of this fungus include high molecular weights and help strengthen the immune system. Furthermore, these polysaccharides act as antioxidants by inhibiting free radicals and enhancing activity of the enzymes. Addition of various elicitors to the fungi submerged culture media affects the cell growth and metabolite production. Fungal extracts are one of these elicitors.
Material and Methods:
In this study, Ganoderma lucidum was first cultured in various culture media to investigate the base media. Using three various methods (soaking the fruit body in water, disintegrating the fruit body with a blender and boiling the fruit body), Agaricus bisporus fruit body extract was prepared as elicitor and the extract with the highest sugar content was used. For the optimization of growth and antioxidant activity of the intracellular polysaccharides, effects of six independent factors were investigated using Placket-Burman method, including Agaricus bisporus extract, peptone, maltose, pH, vitamin B1 and CaCl2. Response surface method was used to optimize three factors of vitamin B1, Agaricus bisporus extract and maltose. Then, stirred tank bioreactor was used to culture Ganoderma lucidum.
Results and Conclusion:
The YPG culture medium was selected as the base medium based on mycelial growth and antioxidant activity of the intracellular polysaccharides (IC50). Sugar content of the Agaricus bisporus extract was 30.66 µg.ml-1. Placket-Burman method revealed that the extracts of Agaricus bisporus, maltose and vitamin B1 significantly increased antioxidant activity of the intracellular polysaccharides. After optimizing these factors using RSM, the IC50 was reported as 1.047 mg.ml-1. Ganoderma lucidum cultivation in bioreactor significantly increased the cell growth (5.29 g.l-1). Intracellular polysaccharides included an IC50 of 1.14 mg.ml-1, which was significantly higher than that the intracellular polysaccharides included in YPG culture media.
Conflict of interest: The authors declare no conflict of interest.
- Agaricus bisporus, Antioxidant activity, Elicitors, Ganoderma lucidum
How to Cite
References
Rathee S, Rathee D, Rathee D, Kumar V,Rathee P, Mushrooms as therapeutic agents. Rev. bras. farmacogn., 2012. 22(2): 459-474.
Dufossé L, Fouillaud M,Caro Y, Fungi and Fungal Metabolites for the Improvement of Human and Animal Nutrition and Health. 2021, Multidisciplinary Digital Publishing Institute.
Alvandi H, Ghahremani M, Hatamian-Zarmi A, Ebrahimi Hosseinzadeh B, Mokhtari-Hosseini Z B,Jafari Farjam S N, Optimization of Soy-based Media for the Production of Biologically Active Exopolysaccharides by Medicinal Mushroom Trametes versicolor. Appl. Food Biotechnol., 2020. 7(4): 251-261.
Alvandi H, Hatamian-Zarmi A, Ebrahimi Hosseinzadeh B,Mokhtari-Hosseini Z-B, Optimization of Production Conditions for Bioactive Polysaccharides from Fomes fomentarius and Investigation of Antibacterial and Antitumor Activities. Iranian Journal of Medical Microbiology, 2020. 14(6): 596-611.
Paterson R R M, Ganoderma–a therapeutic fungal biofactory. Phytochemistry, 2006. 67(18): 1985-2001.
Ahmad M F, Ganoderma lucidum: a rational pharmacological approach to surmount the cancer. Journal of Ethnopharmacology, 2020: 113047.
XiaoPing C, Yan C, ShuiBing L, YouGuo C, JianYun L,LanPing L, Free radical scavenging of Ganoderma lucidum polysaccharides and its effect on antioxidant enzymes and immunity activities in cervical carcinoma rats. Carbohydrate Polymers, 2009. 77(2): 389-393.
YouGuo C, ZongJi S,XiaoPing C, Modulatory effect of Ganoderma lucidum polysaccharides on serum antioxidant enzymes activities in ovarian cancer rats. Carbohydrate Polymers, 2009. 78(2): 258-262.
Arora D S,Chandra P, Assay of antioxidant potential of two Aspergillus isolates by different methods under various physio-chemical conditions. Brazilian Journal of Microbiology, 2010. 41(3): 765-777.
Heydarian M, Hatamian-Zarmi A, Amoabediny G, Ebrahimi-Hosseinzadeh B, Alvandi H, Doryab A,Salehi A, Growth Kinetics and Ganoderic Acid Production from Ganoderma lucidum GIRAN17: A Real-Time Monitoring Platform. Iran-J-Med-Microbiol, 2021. 15(1): 67-84.
Peng L, Qiao S, Xu Z, Guan F, Ding Z, Gu Z, Zhang L,Shi G, Effects of culture conditions on monosaccharide composition of Ganoderma lucidum exopolysaccharide and on activities of related enzymes. Carbohydrate Polymers, 2015. 133: 104-109.
Ai-Lati A, Liu S, Ji Z, Zhang H,Mao J, Structure and bioactivities of a polysaccharide isolated from Ganoderma lucidum in submerged fermentation. Bioengineered, 2017. 8(5): 565-571.
Nojoki F, Hatamian-Zarmi A, Ebrahimi-Hosseinzadeh B, Mir-drikvand M, Mokhtari-Hosseini Z b,Kalantari-Deahaghi S, Investigation and Optimization Effects of ultrasound waves to produce Ganoderic acid, anti-cancer mushrooms metabolite. Iran-J-Med-Microbiol, 2017. 11(1): 58-66.
Ma Z, Xu M, Wang Q, Wang F, Zheng H, Gu Z, Li Y, Shi G,Ding Z, Development of an efficient strategy to improve extracellular polysaccharide production of Ganoderma lucidum using l-phenylalanine as an enhancer. Frontiers in microbiology, 2019. 10: 2306.
Nojoki F, Hatamian-Zarmi A, Mir-Drikvand M, Ebrahimi-Hosseinzadeh B, Mokhtari-Hosseini Z-b, Kalantari-Dehaghi S,Esmaeilifar M, Impact of Rifampin Induction on the Fermentation Production of Ganoderic Acids by Medicinal Mushroom Ganoderma lucidum. Appl. Food Biotechnol., 2016. 3(2): 91-98.
Heydarian M, Hatamian-Zarmi A, Amoabediny G, Yazdian F,Doryab A, Synergistic Effect of Elicitors in Enhancement of Ganoderic Acid Production: Optimization and Gene Expression Studies. Appl. Food Biotechnol., 2015. 2(3): 57-62.
Yuan Y-J, Li C, Hu Z-D,Wu J-C, Signal transduction pathway for oxidative burst and taxol production in suspension cultures of Taxus chinensis var. mairei induced by oligosaccharide from Fusarium oxysprum. Enzyme and microbial technology, 2001. 29(6-7): 372-379.
Zhu L-W, Zhong J-J,Tang Y-J, Significance of fungal elicitors on the production of ganoderic acid and Ganoderma polysaccharides by the submerged culture of medicinal mushroom Ganoderma lucidum. Process Biochemistry, 2008. 43(12): 1359-1370.
Wang W, Yu L,Zhou P, Effects of different fungal elicitors on growth, total carotenoids and astaxanthin formation by Xanthophyllomyces dendrorhous. Bioresource technology, 2006. 97(1): 26-31.
Wasser S P, Current findings, future trends, and unsolved problems in studies of medicinal mushrooms. Applied microbiology and biotechnology, 2011. 89(5): 1323-1332.
Liao S-F, Liang C-H, Ho M-Y, Hsu T-L, Tsai T-I, Hsieh Y S-Y, Tsai C-M, Li S-T, Cheng Y-Y,Tsao S-M, Immunization of fucose-containing polysaccharides from Reishi mushroom induces antibodies to tumor-associated Globo H-series epitopes. Proceedings of the National Academy of Sciences, 2013. 110(34): 13809-13814.
Wang J, Hu S, Nie S, Yu Q,Xie M, Reviews on mechanisms of in vitro antioxidant activity of polysaccharides. Oxidative Medicine and Cellular Longevity, 2016. 2016.
Tang Y-J,Zhong J-J, Fed-batch fermentation of Ganoderma lucidum for hyperproduction of polysaccharide and ganoderic acid. Enzyme and Microbial Technology, 2002. 31(1-2): 20-28.
Masuko T, Minami A, Iwasaki N, Majima T, Nishimura S-I,Lee Y C, Carbohydrate analysis by a phenol–sulfuric acid method in microplate format. Analytical biochemistry, 2005. 339(1): 69-72.
Sharma O P,Bhat T K, DPPH antioxidant assay revisited. Food chemistry, 2009. 113(4): 1202-1205.
Qinnghe C, Xiaoyu Y, Tiangui N, Cheng J,Qiugang M, The screening of culture condition and properties of xylanase by white-rot fungus Pleurotus ostreatus. Process Biochemistry, 2004. 39(11): 1561-1566.
Ouedraogo N, Savadogo A, Somda M K, Tapsoba F, Zongo C,Traore A S, Effect of mineral salts and nitrogen source on yeast (Candida utilis NOY1) biomass production using tubers wastes. African Journal of Biotechnology, 2017. 16(8): 359-365.
Izmirlioglu G,Demirci A, Enhanced bio-ethanol production from industrial potato waste by statistical medium optimization. International journal of molecular sciences, 2015. 16(10): 24490-24505.
Lu J, He R, Sun P, Zhang F, Linhardt R J,Zhang A, Molecular mechanisms of bioactive polysaccharides from Ganoderma lucidum (Lingzhi), a review. International journal of biological macromolecules, 2020. 150: 765-774.
Shi M, Zhang Z,Yang Y, Antioxidant and immunoregulatory activity of Ganoderma lucidum polysaccharide (GLP). Carbohydrate polymers, 2013. 95(1): 200-206.
Qilong S, Effect of different temperature on physiological character-istics of Agaricus bisporus [J]. Science and Technology of Food Industry, 2005. 3.
Alvandi H, Hatamian-Zarmi A, Ebrahimi Hosseinzadeh B, Mokhtari-Hosseini Z-B,Aghajani H, Effect of Production, Extraction and Purification methods on Anti-cancer property of Fungal Polysaccharides. Developmental Biology, 2020. 12(1): 27-42.
Hwang H, Kim S, Xu C, Choi J,Yun J, Production and molecular characteristics of four groups of exopolysaccharides from submerged culture of Phellinus gilvus. Journal of applied microbiology, 2003. 94(4): 708-719.
Boubakri H, Poutaraud A, Wahab M A, Clayeux C, Baltenweck-Guyot R, Steyer D, Marcic C, Mliki A,Soustre-Gacougnolle I, Thiamine modulates metabolism of the phenylpropanoid pathway leading to enhanced resistance to Plasmopara viticola in grapevine. BMC plant biology, 2013. 13(1): 1-15.
Meir Z,Osherov N, Vitamin biosynthesis as an antifungal target. Journal of Fungi, 2018. 4(2): 72.
Kan Y, Chen T, Wu Y,Wu J, Antioxidant activity of polysaccharide extracted from Ganoderma lucidum using response surface methodology. International journal of biological macromolecules, 2015. 72: 151-157.
Tang Y-J, Zhu L-W, Li H-M,Li D-S, Submerged Culture of Mushrooms in Bioreactors--Challenges, Current State-of-the-Art, and Future Prospects. Food Technology & Biotechnology, 2007. 45(3).
Berovič M,Popovic M, Submerged cultivation of Ganoderma lucidum biomass in stirred tank reactor. Chemical and Biochemical Engineering Quarterly, 2018. 32(4): 465-472.
Cao J, Zhang H-J,Xu C-P, Culture characterization of exopolysaccharides with antioxidant activity produced by Pycnoporus sanguineus in stirred-tank and airlift reactors. Journal of the Taiwan Institute of Chemical Engineers, 2014. 45(5): 2075-2080.
Smânia A, Delle Monache F, Smânia E, Gil M, Benchetrit L,Cruz F, Antibacterial activity of a substance produced by the fungus Pycnoporus sanguineus (Fr.) Murr. Journal of ethnopharmacology, 1995. 45(3): 177-181.
- Abstract Viewed: 582 times
- pdf Downloaded: 541 times
