Natural Melanin Synthesized by Aureobasidium pullulans Using Food Wastes and its Characterization Characterization of natural melanin produces by Aureobasidium pullulans
Applied Food Biotechnology,
Vol. 8 No. 4 (2021),
2 Mehr 2021
,
Page 307-318
https://doi.org/10.22037/afb.v8i4.34599
Abstract
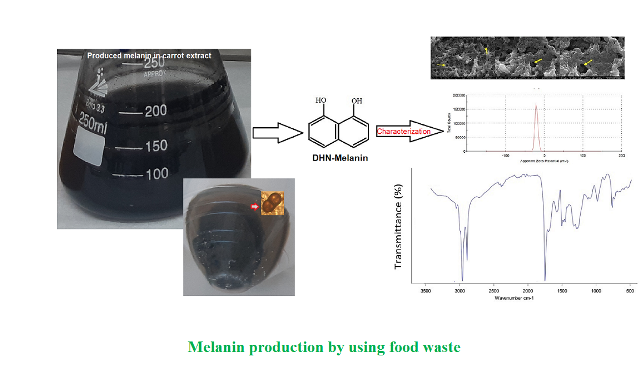
Background and Objective:
Food wastes cause economic losses and environmental problems. Hence, ability to transform food wastes into high-value added products is highly attractive. The aim of this study was to produce melanin pigments by fermentation that include wide potential uses in agriculture, cosmetics and pharmaceutical industries using domestic wastes such as melon peel, watermelon peel and carrot peel and industrial by-products such as whey and molasses.
Material and Methods:
Two Aureobasidium pullulans strains were assessed for melanin production. Fourier-transform infrared spectroscopy, scanning electron microscope, zeta potential, ultraviolet absorbance and solubility assays were carried out to characterize produced melanin nanoparticles.
Results and Conclusion:
The highest intracellular (0.19 g l-1) and extracellular (3.52 g l-1) melanin concentrations were produced by Aureobasidium pullulans NBRC 100716 using carrot peel extracts as fermentation media. Results of characterization were compared with those of synthetic melanin used as standard and the produced nanoparticles were validated. Particle sizes of the nanoparticles ranged 10-760 nm with negative charges, as suggested by previous literature. Results showed that carrot peel was a good candidate, which could be used for the production of high value-added melanin. When carrot peel extract was used as a fermentation medium, characteristics of the melanin produced by Aureobasidium pullulans NBRC 100716 strain were similar to those of synthetic melanin.
Conflict of interest: The authors declare no conflict of interest
- Aureobasidium pullulans; characterisation; food waste; melanin
How to Cite
References
Pralea IE, Moldovan RC, Petrache AM, Ilieș M, Hegheș SC, Lelciu I. From extraction to advanced analytical methods: The challenges of melanin analysis. Int J Mol Sci. 2019; 20:1-37.
doi: 10.3390/ijms201639432.
Almeida-Paes R, Frases S, de Sousa Araujo G, Evangelista de Oliveira MM, Gerfen GJ, Nosanchuk JD. Biosynthesis and functions of a melanoid pigment produced by species of the sporothrix complex in the presence of L-Tyrosine. Appl Environ Microbiol. 2012; 78: 8623-8630.
doi: 10.1128/AEM.02414-12
Liu F, Zhang J, Zhang L, Diao M, Ling P, Wang F. Correlation between the synthesis of pullulan and melanin in Aureobasidium pullulans. Int J Biol Macromol. 2021; 177: 252-260.
doi: 10.1016/j.ijbiomac.2021.02.108
Pombeiro-Sponchiado SR, Sousa GS andrade JCR, Lisboa HF, Gonçalves RCR. Production of Melanin Pigment by Fungi and Its Biotechnological Applications. In: Blumenberg M, Editor. Mela-nin. 2017. Pp. 47-75
Camacho E, Vij R, Chrissian C, Prados-Rosales R, Gil D, O’Meally RN. The structural unit of melanin in the cell wall of the fungal pathogen Cryptococcus neoformans. J Biol Chem. 2019; 294: 10471-10489.
doi: 10.1074/jbc.RA119.008684
Chu M, Hai W, Zhang Z, Wo F, Wu Q, Zhang Z. Melanin nanoparticles derived from a homology of medicine and food for sentinel lymph node mapping and photothermal in vivo cancer therapy. Biomater. 2016; 91:182-199.
doi: 10.1016/j.biomaterials.2016.03.018
Agboyibor C, Kong WB, Chen D, Zhang AM, Niu SQ. Monascus pigments production, composition, bioactivity and its application: A review. Biocatal Agric Biotechnol. 2018; 16: 433-447.
doi: 10.1016/j.bcab.2018.09.012
Lopes FC, Ligabue-Braun R. Agro-industrial residues: Eco-friendly and inexpensive substrates for microbial pigments produc-tion. Fron Sustain Food Syst. 2021; 5: 65.
doi: 10.3389/fsufs.2021.589414
Santhanalakshmi K, Natarajan E, Muthukumar B, Dhanasekaran D, Archunan G. Fermentative production of melanin pigment from Streptomyces griseorubens DKR4 from agro waste products. Int J Appl Res. 2017; 3:284-288.
Roy S, Rhim JW. New insight into melanin for food packaging and biotechnology applications. Crit Rev Food Sci Nutr. 2021; 1-27.
doi: 10.1080/10408398.2021.1878097
El-Gamal MS, El-Bialy HA, Elsayed MA, Khalifa MA. Isolation and characterization of melanized yeast form of Aureobasidium pullulans and physiological studies on the melanization process. J Nucl Technol Appl Sci. 2017; 5:57-72.
Lopusiewicz L. The isolation, purification and analysis of the melanin pigment extracted from Armillaria mellea rhizomorphs. Wolrd Sci News. 2018; 100:135-153.
Panesar R, Kaur S, Panesar PS. Production of microbial pigments utilizing agro-industrial waste: A review. Curr Opin Food Sci. 2015; 1: 70-76.
doi: 10.1016/j.cofs.2014.12.002
Tarangini K, Mishra S. Production of melanin by soil microbial isolate on fruit waste extract: Two step optimization of key parameters. Biotechnol Reports. 2014; 4:139-146.
doi: 10.1016/j.btre.2014.10.001
Mujdeci G, Arevalo-Villena M, Ozbas ZY, Briones Perez A. Yeast identification during fermentation of Turkish gemlik olives. J Food Sci. 2018; 83:1321-1325.
doi: 10.1111/1750-3841.14124
Israilides CJ, Smith A, Harthill JE, Barnett C, Bambalov G, Scanlon B. Pullulan content of the ethanol precipitate from fermented agro- industrial wastes. Appl Microbiol Biotechnol. 1998; 49:613-617.
doi: 10.1007/s002530051222
Mujdeci GN. Investigation of various parameters affecting on pullulan production by Aureobasidium pullulans. 2019: 50.
Suwannarach N, Kumla J, Watanabe B, Matsui K, Lumyong S. Characterization of melanin and optimal conditions for pigment production by an endophytic fungus, Spissiomyces endophytica SDBR-CMU319. Plos One. 2019; 14:1-15.
doi: 10.1371/journal.pone.0222187
Wang H, Pan Y, Tang X, Huang Z. Isolation and characterization of melanin from Osmanthus fragrans’ seeds. LWT Food Sci Technol. 2006; 39:496-502.
doi: 10.1016/j.lwt.2005.04.001
Guo J, Rao Z, Yang T, Man Z, Xu M, Zhang X. High-level production of melanin by a novel isolate of Streptomyces kathirae. FEMS Microbiol Lett. 2014; 357:85-91.
doi: 10.1111/1574-6968.12497
Geib E, Gressler M, Viediernikova I, Hillmann F, Jacobsen ID, Nietzsche S. A non-canonical melanin biosynthesis pathway protects Aspergillus terreus conidia from environmental stress. Cell Chem Biol. 2016; 23: 587-597.
doi: 10.1016/j.chembiol.2016.03.014
Pavan ME, Lopez NI, Pettinari MJ. Melanin biosynthesis in bacteria, regulation and production perspectives. Appl Microbiol Biotechnol. 2020; 104:1357-1370.
doi: 10.1007/s00253-019-10245-y
Plonka PM, Grabacka M. Melanin synthesis in microorganisms -biotechnological and medical aspects. Acta Biochim Pol. 2006; 53:429-443.
Eisenman HC, Greer EM, McGrail CW. The role of melanins in melanotic fungi for pathogenesis and environmental survival. Appl Microbiol Biotechnol. 2020; 104:4247-4257.
doi: 10.1007/s00253-020-10532-z
Dong C, Yao Y. Isolation, characterization of melanin derived from Ophiocordyceps sinensis, an entomogenous fungus endemic to the Tibetan plateau. J Biosci Bioeng. 2012; 113: 474-479.
doi: 10.1016/j.jbiosc.2011.12.001
Suryanarayanan TS, Ravishankar JP, Venkatesan G, Murali TS. Characterization of the melanin pigment of a cosmopolitan fungal endophyte. Mycol Res. 2004; 108: 974-978.
doi: 10.1017/S0953756204000619
Kumar CG, Mongolla P, Pombala S, Kamle A, Joseph J. Physicochemical characterization and antioxidant activity of melanin from a novel strain of Aspergillus bridgeri ICTF‐201. Lett Appl Microbiol. 2011; 53: 350-358.
doi: 10.1111/j.1472-765X.2011.03116.x
Correa N, Covarrubias C, Rodas PI, Hermosilla G, Olate VR, Valdes C. Differential antifungal activity of human and Crypto-coccal melanins with structural discrepancies. Front Microbiol. 2017; 8:1-13.
doi: 10.3389/fmicb.2017.01292
Rageh MM, El-Gebaly RH, Abou-Shady H, Amin DG. Melanin nanoparticles (MNPs) provide protection against whole-body ɣ-irradiation in mice via restoration of hematopoietic tissues. Mol Cell Biochem. 2015; 399(1): 59-69.
doi: 10.1007/s11010-014-2232-y
Caldas M, Santos AC, Veiga F, Rebelo R, Reis RL, Correlo VM. Melanin nanoparticles as a promising tool for biomedical applications - a review. Acta Biomater. 2020; 105: 26-43.
doi: 10.1016/j.actbio.2020.01.044
Roy S, Rhim JW. New insight into melanin for food packaging and biotechnology applications. Crit Rev Food Sci Nutr. 2021; 0(0):1-27.
doi: 10.1080/10408398.2021.1878097
Strube OI, Bungeler A, Bremser W. Site-specific in situ synthesis of eumelanin nanoparticles by an enzymatic autoed-position-like process. Biomacromolecules 2015; 16:1608-1613.
doi: 10.1021/acs.biomac.5b00187
Hou R, Liu X, Yan J, Xiang K, Wu X, Lin W. Characterization of natural melanin from: Auricularia auricula and its hepatoprotective effect on acute alcohol liver injury in mice. Food Funct. 2019; 10:1017-1027.
doi: 10.1039/c8fo01624k
Hu WL, Dai DH, Huang GR, Zhang ZD. Isolation and characterization of extracellular melanin produced by Chroogomphus rutilus D447. Am J Food Technol. 2015; 10: 68-77.
doi: 10.3923/ajft.2015.68.77
Manivasagan P, Venkatesan J, Senthilkumar K, Sivakumar K, Kim SK. Isolation and characterization of biologically active melanin from Actinoalloteichus sp. MA-32. Int J Biol Macromol. 2013; 58:263-274
doi: 10.1016/j.ijbiomac.2013.04.041
- Abstract Viewed: 917 times
- pdf Downloaded: 770 times
