A Strategic Review on Use of Polyhydroxyalkanoates as an Immunostimulant in Aquaculture
Applied Food Biotechnology,
Vol. 8 No. 1 (2021),
14 December 2020
,
Page 1-18
https://doi.org/10.22037/afb.v8i1.31255
Abstract
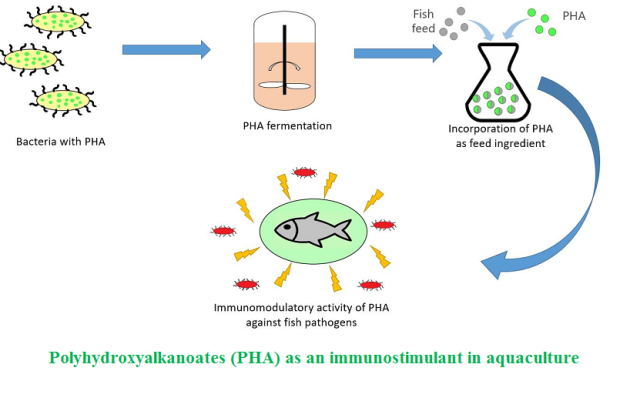
Background and Objective: Increasing concerns over the use of antibiotics in aquaculture have emerged researchers to focus on short chain fatty acids and other biocompatible molecules as alternatives for disease prophylaxis and treatment. Polyhydroxyalkanoates well studied as biopolymeric materials for using in packaging and biomedicine were not focused much for their abilities to act as antimicrobial agents in aquaculture until recent years. Application studies of polyhydroxyalkanoates as aquafeed additives have highlighted their promising roles as eco-friendly alternatives for commercial antibiotics with strong immunomodulatory effects in fish-es and shrimps. The major aim of this review was to explore up-to-date scientific research studies on use of polyhydroxyalkanoates as aquafeed additives and their immunomodulatory effects.
Results and Conclusion: Up-to-date, limited scientific literatures have been published on the use of polyhydroxyalkanoates and their copolymers as alternatives to antibiotics in aquaculture. This research field includes a great scope of development due to the promising immu-nomodulatory and antimicrobial activity of polyhydroxyalkanoates against common pathogens in aquaculture, as reported in literatures. Although several hypothesis and research data for explaining the mechanisms behind their immunostimulatory effects were suggested by various researchers, genetic and molecular bases underlying these phenomena are yet to be explored. Further research and development in this area can introduce these biopolymers as the most promising eco-friendly alternatives for antibiotics in aquaculture.
Conflict of interest: The authors declare no conflict of interest
- Anti-biofilm activity ▪ Agricultural waste ▪ Aquafeed additive ▪ Aquaculture ▪ Immunostimulant ▪ Polyhydroxyalkanoates
How to Cite
References
. Dadar M, Dhama K, Vakharia VN, Hoseinifar SH, Karthik K, Tiwari R, Khandia R, Munjal A, Salgado-Miranda C, Joshi SK. Advances in aquaculture vaccines against fish patho-gens: Global status and current trends. Rev Fish Sci Aquac. 2017; 25(3): 184-217.
doi: 10.1080/23308249.2016.1261277
Pachapur PK, Pachapur VL, Brar SK, Le Bihan Y, Surampalli RY. Food Security and Sustainability. In: Sura-mpalli R, Zhang T, Goyal MK, Brar S, Tyagi R, Editors. Sustainability: Fundamentals and Applications, 1st Edition. John Wiley and Sons, Ltd, 2020; 357-374.
doi: 10.1002/9781119434016.ch17
Little DC, Newton RW, Beveridge MCM. Aquaculture: A rapidly growing and significant source of sustainable food? Status, transitions and potential. Proc Nutr Soc. 2016; 75(3):274-286.
doi: 10.1017/S0029665116000665
FAO, editor. The State of World Fisheries and Aquaculture. Vol. 2020. Food and Agriculture Organization of the United Nations.
doi: 10.4060/ca9229en
Martinez-Porchas M, Martinez-Cordova LR. World aquaculture: Environmental impacts and troubleshooting alternatives. Sci World J. 2012; 1(1):1-9.
doi:10.1100/2012/389623
Biao X, Kaijin Y. Shrimp farming in China: Operating characteristics, environmental impact and perspectives. Ocean Coast Manag. 2007; 50(7):538-550.
doi: 10.1016/j.ocecoaman.2007.02.006
Ma D, Hu Y, Wang J, Ye S, Li A. Effects of antibacterials use in aquaculture on biogeochemical processes in marine sediment. Sci Total Environ. 2006; 367(1):273-277.
doi:10.1016/j.scitotenv.2005.10.014
Ringo E, Olsen RE, Jensen I, Romero J, Lauzon HL. Application of vaccines and dietary supplements in aqua-culture: Possibilities and challenges. Rev Fish Biol Fish. 2014; 24(4): 1005-1032.
doi:10.1007/s11160-014-9361-y
Newman SG. Specific pathogen-free status advances shrimp culture. Global aquaculture advocate. 2009; 12(3):1-4.
Bricknell I, Dalmo RA. The use of immunostimulants in fish larval aquaculture. Fish Shellfish Immunol. 2005; 19(5): 457-472.
doi:10.1016/j.fsi.2005.03.008
Sakai M. Current research status of fish immunostimulants. Aquaculture 1999; 172(1):63-92.
doi: 10.1016/S0044-8486(98)00436-0
Crab R, Defoirdt T, Bossier P, Verstraete W. Biofloc technology in aquaculture: beneficial effects and future challenges. Aquaculture 2012; 356-357:351-356.
doi: 10.1016/j.aquaculture.2012.04.046
Hoseinifar SH, Sun Y-Z, Caipang CM. Short-chain fatty acids as feed supplements for sustainable aquaculture: An updated view. Aquac Res. 2017; 48(4):1380-1391.
doi: 10.1111/are.13239
Defoirdt T, Boon N, Sorgeloos P, Verstraete W, Bossier P. Alternatives to antibiotics to control bacterial infections: Luminescent vibriosis in aquaculture as an example. Trends Biotechnol. 2007; 25(10): 472-479.
doi:10.1016/j.tibtech.2007.08.001
Rajeswari V, Priyadarshini SK, Saranya V, Suguna P, Shenbagarathai R. Immunostimulation by phospholipopeptide biosurfactant from Staphylococcus hominis in Oreochromis mossambicus. Fish Shellfish Immun. 2016; 48: 244-253.
doi:10.1016/j.fsi.2015.11.006
Giri SS, Chi C, Jun JW, Park SC. Use of bacterial subcellular components as immunostimulants in fish aquaculture. Rev Aquacult. 2018; 10(2):474-492.
doi:10.1111/raq.12182
Duan Y, Zhang Y, Dong H, Zheng X, Wang Y, Li H, Liu Q, Zhang J. Effect of dietary poly-β-hydroxybutyrate (PHB) on growth performance, intestinal health status and body composition of Pacific white shrimp Litopenaeus vannamei (Boone, 1931). Fish Shellfish Immunol. 2017; 60:520-528. doi:0.1016/j.fsi.2016.11.020
Anderson AJ, Dawes EA. Occurrence, metabolism, metabolic role, and industrial uses of bacterial polyhydroxy-alkanoates. Microbiol Rev. 1990; 54(4): 450-472.
doi: 10.1128/MMBR.54.4.450-472.1990
Obruca S, Sedlacek P, Slaninova E, Fritz I, Daffert C, Meixner K, Sedrlova Z, Koller M. Novel unexpected functions of PHA granules. Appl Microbiol Biotechnol. 2020; 104(11): 4795-4810.
doi:10.1007/s00253-020-10568-1
Khosravi-Darani K, Bucci DZ. Application of poly (hydroxyalkanoate) in food packaging: Improvements by nanotechnology. Chem Biochem Eng Q. 2015; 29(2):275-285.
doi: 10.15255/CABEQ.2014.2260
Koller M. Poly (hydroxyalkanoates) for food packaging: Application and attempts towards implementation.Apple food Biotechnol. 2014; 1(1):3-15.
doi: 10.22037/afb.v1i1.7127
Chee JY, Lakshmanan M, Jeepery IF, Hairudin NHM, Sudesh K. The potential application of Cupriavidus necator as polyhydroxyalkanoates producer and single cell protein: A review on scientific, cultural and religious perspectives. Appl Food Biotechnol. 2019; 6(1): 19-34.
doi: 10.22037/afb.v6i1.22234
Umesh M, Priyanka K, Thazeem B, Preethi K. Biogenic PHA nanoparticle synthesis and characterization from Bacillus subtilis NCDC0671 using orange peel medium. 2018; 67(17): 996-1004.
doi:10.1080/00914037.2017.1417284
Umesh M, Thazeem B. Biodegradation studies of polyhydroxyalkanoates extracted from Bacillus subtilis NCDC 0671. Res J Chem Environ. 2019; 23(6):107-114.
Koller M, Marsalek L, de Sousa Dias MM, Braunegg G. Producing microbial polyhydroxyalkanoate (PHA) bio-polyesters in a sustainable manner. N Biotechnol. 2017; 37(1):24-38.
doi: 10.1016/j.nbt.2016.05.001
Koller M, Braunegg G. Potential and prospects of continuous polyhydroxyalkanoate (pha) production. Bio-engineering (basel). 2015; 2(2):94-121.
doi: 10.3390/bioengineering2020094
Koller M, Marsalek L. Potential of diverse prokaryotic organisms for glycerol-based Polyhydroxyalkanoate pro-duction. Appl Food Biotechnol. 2015; 2(3):3-15.
doi:10.22037/afb.v2i3.8271
Koller M, Dias MM de S, Rodriguez-Contreras A, Kunaver M, Zagar E, Krzan A, Braunegg G. Liquefied wood as inexpensive precursor-feedstock for bio-mediated incorporation of (R)-3-hydroxyvalerate into polyhydro-xyalkanoates. Mater. 2015; 8(9):6543-6557.
doi:10.3390/ma8095321
Priyanka K, Umesh M, Thazeem B, Preethi K. Polyhydroxyalkanoate biosynthesis and characterization from optimized medium utilizing distillery effluent using Bacillus endophyticus MTCC 9021: A statistical approach. Biocatal Biotransfor. 2020;1(1):1-13.
Khosravi DK, Vasheghani FE, Tanaka K. Hydrogen oxidizing bacteria as poly (hydroxybutyrate) produ-cers. Iranian J Biotechnol. 2006; 4(3):193-196.
Khosravi-Darani K, Mokhtari Z-B, Amai T, Tanaka K. Microbial production of poly(hydroxybutyrate) from C1 carbon sources. Appl Microbiol Biotechnol. 2013; 97(4): 1407-1424.
doi:10.1007/s00253-012-4649-0
Khosravi-Darani K, Yazdian F, Babapour F, Amirsadeghi AR. Poly (3-hydroxybutyrate) production from natural gas by a methanotroph native bacterium in a bubble column bioreactor. Chem Biochem Eng Q. 2019; 33(1):69-77.
doi: 10.15255/CABEQ.2017.1263
Ghoddosi F, Golzar H, Yazdian F, Khosravi-Darani K, Vasheghani-Farahani E. Effect of carbon sources for PHB production in bubble column bioreactor: Emphasis on imp-rovement of methane uptake. J Environ Chem Eng. 2019; 7(2):1-6.
doi: 10.1016/j.jece.2019.102978
Mokhtari-Hosseini ZB, Vasheghani-Farahani E, Heida-rzadeh-Vazifekhoran A, Shojaosadati SA, Karimzadeh R, Khosravi Darani K. Statistical media optimization for growth and PHB production from methanol by a methyl-otrophic bacterium. Bioresour Technol. 2009; 100(8):2436-2443.
doi:10.1016/j.biortech.2008.11.024
Mokhtari-Hosseini ZB, Vasheghani-Farahani E, Shoja-osadati SA, Karimzadeh R, Heidarzadeh-Vazifekhoran A. Effect of feed composition on PHB production from methanol by HCDC of Methylobacterium extorquens (DS-MZ 1340). J Chem Technol Biotechnol
. 2009; 84(8):1136-1139.
Shahhosseini S, Khosravi DK. Simulation and model validation of batch PHB production process using Ralstonia eutropha. Iran J Chem Chem Eng. 2003; 22(2): 35-42.
Darani K, Farahani KVE, Shojaosadati SA. Application of the plackett-burman design for the optimization of poly (β-Hydroxybutyrate) production by Ralstonia eutropha. Iran J Biotechnol. 2003; 1: 155.
Khosravi DK, Vasheghani FE, Shoja ASA. Application of the Taguchi design for production of poly (β-hydroxy-butyrate) by Ralstonia eutropha. Iran J Chem Chem Eng. 2004; 23(1):131-136.
Khosravi-Darani K, Vasheghani-Farahani E. Application of supercritical fluid extraction in biotechnology. Crit Rev Biotechnol. 2005; 25(4): 231-242.
doi:10.1080/07388550500354841
Gao M, Du D, Bo Z, Sui L. Poly-β-hydroxybutyrate (PHB)-accumulating Halomonas improves the survival, growth, robustness and modifies the gut microbial composition of Litopenaeus vannamei postlarvae. Aquaculture 2019;
(1):607-612.
doi: 10.1016/j.aquaculture.2018.10.032
Suguna P, Binuramesh C, Abirami P, Saranya V, Poornima K, Rajeswari V, Shenbagarathai R. Immunostimulation by poly-β hydroxybutyrate–hydroxyvalerate (PHB–HV) from Bacillus thuringiensis in Oreochromis mossambicus. Fish Shellfish Immun. 2014; 36(1): 90-97.
doi: 10.1016/j.fsi.2013.10.012
Tan G-Y, Chen C-L, Li L, Ge L, Wang L, Razaad I, Li Y, Zhao L, Mo Y, Wang J-Y. Start a research on biopolymer polyhydroxyalkanoate (PHA): A review. Polyme. 2014; 6(3): 706-754.
do:10.3390/polym6030706
Jung YM, Lee YH. Utilization of oxidative pressure for enhanced production of poly-beta-hydroxybutyrate and poly (3-hydroxybutyrate-3-hydroxyvalerate) in Ralstonia eutropha. J Biosci Bioeng. 2000; 90(3):266-270.
doi: 10.1016/S1389-1723(00)80080-8
Naik S, Venu Gopal SK, Somal P. Bioproduction of polyhydroxyalkanoates from bacteria: a metabolic appro-ach. World J Microbiol Biotechnol. 2008; 24(10): 2307-2314.
doi:10.1007/s11274-008-9745-z
Liebergesell M, Mayer F, Steinbuchel A. Anaylsis of polyhydroxyalkanoic acid-biosynthesis genes of anoxy-genic phototrophic bacteria reveals synthesis of a polyester exhibiting an unusal composition. Appl Microbiol Bio-technol. 1993; 40(1): 292-300.
doi:10.1007/bf00170383
Tsuge T, Yano K, Imazu S-I, Numata K, Kikkawa Y, Abe H, Taguchi S, Doi Y. Biosynthesis of polyhydroxyalkanoate (PHA) copolymer from fructose using wild-type and laboratory-evolved PHA synthases. Macromol Biosci. 2005; 5(2):112-117.
doi:10.1002/mabi.200400152
Luengo JM, Garcia B, Sandoval A, Naharro G, Olivera ER. Bioplastics from microorganisms. Curr Opin Microbiol. 2003; 6(3): 251-260.
doi: 10.1016/S1369-5274(03)00040-7
Martinez-Tobon DI, Gul M, Elias AL, Sauvageau D. Polyhydroxybutyrate (PHB) biodegradation using bacterial strains with demonstrated and predicted PHB depolymerase activity. Appl Microbiol Biotechnol. 2018; 102(18):8049-8067.
doi: 10.1007/s00253-018-9153-8
Prieto A, Escapa IF, Martinez V, Dinjaski N, Herencias C, de la Pena F, Tarazona N, Revelles O. A holistic view of polyhydroxyalkanoate metabolism in Pseudomonas putida. Environ Microbiol. 2016; 18(2):341-357.
doi:10.1111/1462-2920.12760
Hiraishi T, Taguchi S. Protein engineering of enzymes involved in bioplastic metabolism. In: Ogawa t, protein engineering-technology and application. 1st edition. InTech, London, 2013:133-165.
doi: doi.org/10.5772/55552
Ong SY, Chee JY, Sudesh K. Degradation of poly-hydroxyalkanoate (PHA): A review. J Sib Fed Univ, Biol. 2017; 10(2):211-225.
doi: 10.17516/1997-1389-0024
Kobayashi T, Uchino K, Abe T, Yamazaki Y, Saito T. Novel intracellular 3-hydroxybutyrate-oligomer hydrolase in Wautersia eutropha H16. J Bacteriol. 2005; 187(15): 5129-5135.
doi:10.1128/JB.187.15.5129-5135.2005
Lemes AP, Montanheiro TLA, Passador FR, Duran N. Nanocomposites of Polyhydroxyalkanoates Reinforced with Carbon Nanotubes: Chemical and Biological Pro-perties. In: Thakur VK, Thakur MK, Eco-Friendly Polymer Nanocomposites: Processing and Properties. 1st Edition. Springer, New Delhi, India; 2015:79-108.
doi:. 10.1007/978-81-322-2470-9_3
Blevins HM, Blue MKD, Cobbs BD, Ricotilli TA. Characterization of an extracellular polyhydroxyalkanoate depolymerase from Streptomyces sp. SFB5A. J Bioremediat Biodegrad. 2018; 9(5):1-11
doi: 10.4172/2155-6199.1000452
Kheti B, Kamilya D, Choudhury J, Parhi J, Debbarma M, Singh ST. Dietary microbial floc potentiates immune response, immune relevant gene expression and disease res-istance in rohu, Labeo rohita (Hamilton, 1822) fingerlings. Aquaculture 2017; 468: 501-507.
doi: 10.1016/j.aquaculture.2016.11.018
Sui L, Liu Y, Sun H, Wille M, Bossier P, De Schryver P. The effect of poly-β-hydroxybutyrate on the performance of Chinese mitten crab (Eriocheir sinensis Milne-Edwards) zoea larvae. Aquac Res. 2014; 45(3):558-565.
doi: 10.1111/are.12077
Yaqoob T, Khan N, Arslan M, Korkmaz F, Tacer A, Suzer C, Dogar S. Dietary Supplementation of Poly-AA2-Hydro-xybutyrate on the Growth, Digestive Enzymes Activity and Body Composition of Rainbow Trout (Onchorynchus mykiss). Res Rev J Zool Sci. 2018;6(1):38–42.
Nhan DT, Dinh The Nhan, Wille M, De Schryver P, Defoirdt T, Bossier P, Sorgeloos P. The effect of poly β-hydroxybutyrate on larviculture of the giant freshwater prawn Macrobrachium rosenbergii. Aquaculture. 2010; 302(1-2):76-81.
doi:10.1016/j.aquaculture.2010.02.011
Defoirdt T, Halet D, Vervaeren H, Boon N, Van de Wiele T, Sorgeloos P, Bossier P, Verstraete W. The bacterial storage compound poly-beta-hydroxybutyrate protects Artemia franciscana from pathogenic Vibrio campbellii. Environ Microbiol. 2007; 9(2): 445-452.
doi:10.1111/j.1462-2920.2006.01161.x
De Schryver P, Sinha AK, Kunwar PS, Baruah K, Verstraete W, Boon N, De Boeck G, Bossier P. Poly-beta-hydroxy-butyrate (PHB) increases growth performance and intestinal bacterial range-weighted richness in juvenile European sea bass, Dicentrarchus labrax. Appl Microbiol Biotechnol. 2010; 86(5): 1535-1541.
doi: 10.1007/s00253-009-2414-9
Wang X, Jiang X, Wu F, Ma Y, Che X, Chen X, Liu P, Zhang W, Ma X, Chen G. Microbial poly‐3‐hydroxy-butyrate (PHB) as a feed additive for fishes and piglets. Biotechnol J. 2019; 14(12): 1-8.
doi: 10.1002/biot.201900132
Situmorang ML, De Schryver P, Dierckens K, Bossier P. Effect of poly-β-hydroxybutyrate on growth and disease resistance of Nile tilapia Oreochromis niloticus juveniles. Vet Microbiol. 2016; 182:44-49.
doi: 10.1016/j.vetmic.2015.10.024
Castro R, Jouneau L, Tacchi L, Macqueen DJ, Alzaid A, Secombes CJ, Martin SAM, Boudinot P. Disparate developmental patterns of immune responses to bacterial and viral infections in fish. Sci Rep. 2015; 5:1-12.
doi: 10.1038/srep15458
Franke A, Roth O, Schryver PD, Bayer T, Garcia-Gonzalez L, Künzel S, Bossier P, Miest JJ, Clemmesen C. Poly-β-hydroxybutyrate administration during early life: Effects on performance, immunity and microbial community of European sea bass yolk-sac larvae. Sci Rep. 2017; 7(1): 1-11.
doi: 10.1038/s41598-017-14785-z
Franke A, Clemmesen C, De Schryver P, Garcia-Gonzalez L, Miest JJ, Roth O. Immunostimulatory effects of dietary poly-β-hydroxybutyrate in European sea bass post larvae. Aquac Res. 2017; 48(12): 5707-5717.
doi: 10.1111/are.13393
Najdegerami EH, Tran TN, Defoirdt T, Marzorati M, Sorgeloos P, Boon N, Bossier P. Effects of poly-β-hydroxy-butyrate (PHB) on Siberian sturgeon (Acipenser baerii) fingerlings performance and its gastrointestinal tract microbial community. FEMS Microbiol Ecol. 2017; 79(1): 25-33.
doi: 10.1111/j.1574-6941.2011.01194.x
Najdegerami EH, Baruah K, Shiri A, Rekecki A, Van den Broeck W, Sorgeloos P, Boon N, Bossier P, De Schryver P. Siberian sturgeon (Acipenser baerii) larvae fed Artemia nauplii enriched with poly-β-hydroxybutyrate (PHB): Effect on growth performance, body composition, digestive enzymes, gut microbial community, gut histology and stress tests. Aquac Res. 2015; 46(4): 801-812.
doi: 10.1111/are.12231
Misra S, Sahu NP, Pal AK, Xavier B, Kumar S, Mukherjee SC. Pre and post-challenge immuno-haematological changes in Labeo rohita juveniles fed gelatinised or non-gelatinised carbohydrate with n-3 PUFA. Fish Shellfish Immunol. 2006; 21(4): 346-356.
doi: 10.1016/j.fsi.2005.12.010
Alexander JB, Ingram GA. Noncellular nonspecific defence mechanisms of fish. Annu Rev Fish Dis. 1992; 2:249-279.
doi: 10.1016/0959-8030(92)90066-7
Fu GH, Bai ZY, Xia JH, Liu F, Liu P, Yue GH. Analysis of two lysozyme genes and antimicrobial functions of their recombinant proteins in Asian seabass. Plos One. 2013; 8:1-12.
doi: 10.1371/journal.pone.0079743
Clements KD, Gleeson VP, Slaytor M. Short-chain fatty acid metabolism in temperate marine herbivorous fish. J Comp Physiol B. 1994; 164: 372-327.
doi: 10.1007/bf00302552
Laranja JLQ, Amar EC, Ludevese-Pascual GL, Niu Y, Geaga MJ, De Schryver P, Bossier P. A probiotic Bacillus strain containing amorphous poly-beta-hydroxybutyrate (PHB) stimulates the innate immune response of Penaeus monodon postlarvae. Fish Shellfish Immunol. 2017; 68:202-210.
doi: 10.1016/j.fsi.2017.07.023
Amparyup P, Charoensapsri W, Tassanakajon A. Prophenoloxidase system and its role in shrimp immune responses against major pathogens. Fish Shellfish Immunol. 2013; 34(4): 990-1001.
doi:10.1016/j.fsi.2012.08.019
Maningas MBB, Kondo H, Hirono I, Saito-Taki T, Aoki T. Essential function of transglutaminase and clotting protein in shrimp immunity. Mol Immunol. 2008; 45(5): 1269-1275.
doi: 10.1016/j.molimm.2007.09.016
Morimoto RI. Regulation of the heat shock transcriptional response: Cross talk between a family of heat shock factors, molecular chaperones, and negative regulators. Genes Dev. 1998; 12(24): 3788-3796.
doi: 10.1101/gad.12.24.3788
Qiao G, Xu C, Sun Q, Xu D-H, Zhang M, Chen P, Li Q. Effects of dietary poly-β-hydroxybutyrate supplementation on the growth, immune response and intestinal microbiota of soiny mullet (Liza haematocheila). Fish Shellfish Immunol. 2019; 91:251-263.
doi:10.1016/j.fsi.2019.05.038
Ighodaro OM, Akinloye OA. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alexandria J Med. 2018; 54(4):287-293.
doi: 10.1016/j.ajme.2017.09.001
Bagnyukova TV, Vasylkiv OY, Storey KB, Lushchak VI. Catalase inhibition by amino triazole induces oxidative stress in goldfish brain. Brain Res. 2005; 1052(2):180-186.
doi: 10.1016/j.brainres.2005.06.002
Meng Q, Chen J, Xu C, Huang Y, Wang Y, Wang T, Zhai X, Gu W, Wang W. The characterization, expression and activity analysis of superoxide dismutases (SODs) from Procambarus clarkii. Aquaculture. 2013; 406-407:131-140.
doi: 10.1016/j.aquaculture.2013.05.008
Ishida K, Hung TV, Liou K, Lee HC, Shin C-H, Sohng JK. Characterization of pbpA and pbp2 encoding penicillin-binding proteins located on the downstream of clavulanic acid gene cluster in Streptomyces clavuligerus. Biotechnol Lett. 2006; 28(6): 409-417.
doi: 10.1007/s10529-005-6071-5
Fujimoto Y, Konishi Y, Kubo O, Hasegawa M, Inohara N, Fukase K. Synthesis of crosslinked peptidoglycan frag-ments for investigation of their immunobiological functions. Tetrahedron Lett. 2009; 50(26): 3631-3634.
doi: 10.1016/j.tetlet.2009.03.081
Baggiolini M, Dewald B, Moser B. lnterleukin-8 and related chemotactic cytokines-CXC and CC chemokines. Adv Immunol. 1993; 55:97-179.
doi: 10.1016/s0065-2776(08)60509-x
Wright RM, Weigel LK, Varella-Garcia M, Vaitaitis G, Repine JE. Molecular cloning, refined chromosomal mapping and structural analysis of the human gene encoding aldehyde oxidase (AOX1), a candidate for the ALS2 gene. Redox Rep. 1997;3 (3):135-144.
doi:10.1080/13510002.1997.11747101
Ryhanen P, Surcel HM, Ilonen J. Decreased expression of class II major histocompatibility complex (MHC) molecules on monocytes is found in open-heart surgery related immu-nosuppression. Acta Anaesthesiol Scand. 1991; 35(5):453-456.
doi: 10.1111/j.1399-6576. 1991.tb03327.x
Semova I, Carten JD, Stombaugh J, Mackey LC, Knight R, Farber SA, Rawls JF. Microbiota regulate intestinal absorption and metabolism of fatty acids in the zebrafish. Cell Host Microbe. 2012; 12(3):277-288.
doi: 10.1016/j.chom.2012.08.003
Luo G, Liu Z, Shao L, Tan H. Using poly-β-hydroxybutyric as an additional carbohydrate for biofloc in a shrimp Litopenaeus vannamei bioflocs nursery system with brackish water. Aquaculture. 2019; 506: 181-187.
doi: 10.1016/j.aquaculture.2019.03.021
Sui L, Cai J, Sun H, Wille M, Bossier P. Effect of poly-β-hydroxybutyrate on Chinese mitten crab, Eriocheir sinensis, larvae challenged with pathogenic Vibrio anguillarum. J Fish Dis. 2012; 35(5): 359-364.
doi: 10.1111/j.1365-2761.2012. 01351.x
Baruah K, Huy TT, Norouzitallab P, Niu Y, Gupta SK, De Schryver P, Bossier P. Probing the protective mechanism of poly-ß-hydroxybutyrate against vibriosis by using gnoto-biotic Artemia franciscana and Vibrio campbellii as host-pathogen model. Sci Rep. 2015; 5:1-8.
doi: 10.1038/srep09427
Halet D, Defoirdt T, Van Damme P, Vervaeren H, Forrez I, Van de Wiele T, Boon N, Sorgeloos P, Bossier P, Verstraete W. Poly-beta-hydroxybutyrate-accumulating bacteria protect gnotobiotic Artemia franciscana from pathogenic Vibrio campbellii. FEMS Microbiol Ecol. 2007; 60(3): 363-369.
doi: 10.1111/j.1574-6941.2007. 00305.x
Van Hung N, De Schryver P, Tam TT, Garcia-Gonzalez L, Bossier P, Nevejan N. Application of poly-β-hydroxy-butyrate (PHB) in mussel larviculture. Aquaculture 2015; 446:318-324.
doi: 10.1016/j.aquaculture.2015.04.036
Thai TQ, Wille M, Garcia-Gonzalez L, Sorgeloos P, Bossier P, De Schryver P. Poly-ß-hydroxybutyrate content and dose of the bacterial carrier for Artemia enrichment determine the performance of giant freshwater prawn larvae. Appl Microbiol Biotechnol. 2014; 98(11):5205-5215.
doi: 10.1007/s00253-014-5536-7
Najdegerami H E. Immunostimulatory and growth-promo-ting potential of poly-β-hydroxybutyrate in rainbow trout (Oncorhynchus mykiss) fingerlings culture. Iran J Fish Sci. 2020; 19(2): 847-865.
Laranja JLQ, Ludevese-Pascual GL, Amar EC, Sorgeloos P, Bossier P, De Schryver P. Poly-β-hydroxybutyrate (PHB) accumulating Bacillus spp. improve the survival, growth and robustness of Penaeus monodon (Fabricius, 1798) postlarvae. Vet Microbiol. 2014; 173(3-4):310-317.
doi: 10.1016/j.vetmic.2014.08.011
Costerton JW, William Costerton J, Lappin-Scott H. Introduction to Microbial Biofilms. Microbial Biofilms. 1995:1-12.
doi: 10.1017/cbo9780511525353.002
Kiran GS, Lipton AN, Priyadharshini S, Anitha K, Suarez LEC, Arasu MV, Choi KC, Selvin J, Al-Dhabi NA. Antiadhesive activity of poly-hydroxy butyrate biopolymer from a marine Brevibacterium casei MSI04 against shrimp pathogenic vibrios. Microb Cell Fact. 2014; 13:114.
doi: 10.1186/s12934-014-0114-3
Lee CW, Song BK, Jegal J, Kimura Y. Cell adhesion and surface chemistry of biodegradable aliphatic polyesters: Discovery of particularly low cell adhesion behavior on poly(3-[RS]-hydroxybutyrate). Macromolecular Research. 2013; 21:1305-1313.
doi: 10.1007/s13233-013-1181-8
Van Hung N, Bossier P, Hong NTX, Ludeseve C, Garcia‐Gonzalez L, Nevejan N, De Schryver P. Does Ralstonia eutropha, rich in poly‐β hydroxybutyrate (PHB), protect blue mussel larvae against pathogenic vibrios ?. J Fish Dis. 2019; 42(6):777-787.
doi: 10.1111/jfd.12981
Fernandez RG. Artemia bioencapsulation 1. Effect of particle sizes on the filtering behavior of Artemia franciscana. J Crustacean Biol. 2001; 21(2): 435-442.
doi: 10.1163/20021975-99990144
Vogel C, Marcotte EM. Insights into the regulation of protein abundance from proteomic and transcriptomic analyses. Nat Rev Genet. 2012; 13(4):227-232.
doi: 10.1038/nrg3185
Weinberg ED. Iron availability and infection. Bba-Gen Subjects. 2009; 1790(7):600-605
doi: 10.1016/j.bbagen.2008.07.002
Van Hung N, De Schryver P, Dung NV, Nevejan N, Bossier P. Ralstonia eutropha, containing high poly-β-hydroxy-butyrate levels, regulates the immune response in mussel larvae challenged with Vibrio coralliilyticus. Fish Shellfish Immunol. 2019; 84:196-203.
doi: 10.1016/j.fsi.2018.09.066
Preethi K, Vineetha UM. Water hyacinth: A potential substrate for bioplastic (PHA) production using Pseudo-monas aeruginosa. Int J Appl Res Vet Med. 2015; 1(11):349-354.
Yin F, Li D, Ma X, Li J, Qiu Y. Poly (3-hydroxybutyrate-3-hydroxyvalerate) production from pretreated waste lignocellulosic hydrolysates and acetate cosubstrate. Bioresour Technol. 2020; 316: 123911.
doi: 10.1016/j.biortech.2020.123911
Gowda V, Shivakumar S. Agrowaste-based Polyhydroxy-alkanoate (PHA) production using hydrolytic potential of Bacillus thuringiensis IAM 12077. Braz Arch Biol Technol. 2014; 57(1):55-61.
doi: 10.1590/S1516-89132014000100009
Davis R, Kataria R, Cerrone F, Woods T, Kenny S, O’Donovan A, Guzik M, Shaikh H, Duane G, Gupta VK, Tuohy MG, Padamatti RB, Casey E, O’Connor KE. Conversion of grass biomass into fermentable sugars and its utilization for medium chain length polyhydroxyalkanoate (mcl-PHA) production by Pseudomonas strains. Bioresour Technol. 2013; 150:202-209.
doi: 10.1016/j.biortech.2013.10.001
Lee SY. Plastic bacteria? Progress and prospects for polyhyd-roxyalkanoate production in bacteria. Trends Biotechnol. 1996; 14(11):431-438.
Defoirdt T, Boon N, Sorgeloos P, Verstraete W, Bossier P. Short-chain fatty acids and poly-β-hydroxyalkanoates: (New) Biocontrol agents for a sustainable animal prod-uction. Biotechnol Adv. 2009; 27(6):680-685.
doi: 10.1016/j.biotechadv.2009.04.026
Madkour MH, Heinrich D, Alghamdi MA, Shabbaj II, Steinbüchel A. PHA recovery from biomass. Biomacromolecules. 2013; 14(9): 2963-2972.
doi: 10.1021/bm4010244
Kunasundari B, Sudesh K. Isolation and recovery of microbial polyhydroxyalkanoates. Express Polym Lett. 2011; 5(7): 620-634.
doi: 10.3144/expresspolymlett.2011.60
Kourmentza C, Placido J, Venetsaneas N, Burniol-Figols A, Varrone C, Gavala HN, Reis MAM. Recent advances and challenges towards sustainable Polyhydroxyalkanoate (PHA) production. Bioengin. 2017; 4(2)1-43.
doi: 10.3390/bioengineering4020055
Anis SNS, Iqbal NM, Kumar S, Al-Ashraf A. Increased recovery and improved purity of PHA from recombinant Cupriavidus necator. Bioengineered 2013; 4(2): 115-118.
doi: 10.4161/bioe.22350
Neves A, Muller J. Use of enzymes in extraction of polyhydroxyalkanoates produced by Cupriavidus necator. Biotechnol Progr. 2012; 28(6):1575-1580.
doi: 10.1002/btpr.1624
Santimano MC, Prabhu NN, Garg S. PHA Production Using Low-Cost Agro-Industrial Wastes by Bacillus sp. Strain COL1/A6. Res J Microbiol. 2009; 4(3): 89-96.
doi: 10.3923/jm.2009.89.96
Khandpur P, Jabeen ET, Rohini KVL, Varaprasad Y. Study on production, extraction and analysis of polyhydroxy-alkanoate (PHA) from bacterial isolates. IOSR J Pharm Biol Sci. 2012; 1:31-38.
doi: 10.9790/3008-0113138
Van-Thuoc D, Quillaguaman J, Mamo G, Mattiasson B. Utilization of agricultural residues for poly(3-hydroxy-butyrate) production by Halomonas boliviensis LC1. J Appl Microbiol. 2008; 104(2): 420-428.
do:10.1111/j.1365-2672.2007. 03553.x
Kulkarni SO, Kanekar PP, Jog JP, Sarnaik SS, Nilegaonkar SS. Production of copolymer, poly (hydroxybutyrate-co-hydroxyvalerate) by Halomonas campisalis MCM B-1027 using agro-wastes. Int J Biol Macromol. 2015; 72:784-789.
doi: 10.1016/j.ijbiomac.2014.09.028
Chaudhry WN, Jamil N, Ali I, Ayaz MH, Hasnain S. Screening for polyhydroxyalkanoate (PHA)- producing bacterial strains and comparison of PHA production from various inexpensive carbon sources. Ann Microbiol. 2011; 61(3): 623-629.
doi: 10.1007/s13213-010-0181-6
Queiros D, Rossetti S, Serafim LS. PHA production by mixed cultures: a way to valorize wastes from pulp industry. Bioresour Technol. 2014; 157: 197-205.
doi: 10.1016/j.biortech.2014.01.099
Saranya V, Shenbagarathai R. Production and charac-terization of PHA from recombinant E. coli harbouring phaC1 gene of indigenous Pseudomonas sp. LDC-5 using molasses. Braz J Microbiol. 2011; 42(3):1109-1118.
doi: 10.1590/S1517-83822011000300032
Bhuwal AK, Singh G, Aggarwal NK, Goyal V, Yadav A. Isolation and screening of polyhydroxyalkanoates produ-cing bacteria from pulp, paper, and cardboard industry wastes. Int J Biomater. 2013; 2013:1-10.
doi:10.1155/2013/752821
Obruca S, Benesova P, Petrik S, Oborna J, Prikryl R, Marova I. Production of polyhydroxyalkanoates using hydrolysate of spent coffee grounds. Process Biochem. 2014; 49(9): 1409-1414.
doi: 10.1016/j.procbio.2014.05.013
Jiang Y, Marang L, Tamis J, van Loosdrecht MCM, Dijkman H, Kleerebezem R. Waste to resource: Converting paper mill wastewater to bioplastic. Water Res. 2012; 46(17): 5517-5530.
doi: 10.1016/j.watres.2012.07.028
Koller M. Recycling of waste streams of the biotechnological poly (hydroxyalkanoate) production by Haloferax medit-erranei on Whey. Int J Polym Sci. 2015; 2015:1-9.
doi: 10.1155/2015/370164
Elain A, Le Grand A, Corre Y-M, Le Fellic M, Hachet N, Le Tilly V, Loulergue P, Audic J-L, Bruzaud S. Valorisation of local agro-industrial processing waters as growth media for polyhydroxyalkanoates (PHA) production. Ind Crops Prod. 2016; 80:1-5.
doi: 10.1016/j.indcrop.2015.10.052
Anjali M, Sukumar C, Kanakalakshmi A, Shanthi K. Enhancement of growth and production of poly-hydroxyalkanoates by Bacillus subtilis from agro-industrial waste as carbon substrates. Compos Interfaces. 2014; 21(2):111-119.
doi:10.1080/15685543.2013.834200
Vega-Castro O, Contreras-Calderon J, Leon E, Segura A, Arias M, Perez L, Sobral PJA. Characterization of a polyhydroxyalkanoate obtained from pineapple peel waste using Ralsthonia eutropha. J Biotechnol. 2016; 231:232-238.
doi:10.1016/j.jbiotec.2016.06.018
Rodriguez-Carmona E, Bastida J, Manresa A. Utilization of agro-industrial residues for poly(3-hydroxyalkanoate) production by Pseudomonas aeruginosa 42A2 (NCIMB 40045): Optimization of culture medium. J Am Oil Chem Soc. 2012; 89(1):111-122.
doi:10.1007/s11746-011-1897-6
Costa SGVAO, Lepine F, Milot S, Deziel E, Nitschke M, Contiero J. Cassava wastewater as a substrate for the simultaneous production of rhamnolipids and polyhydroxy-alkanoates by Pseudomonas aeruginosa. J Ind Microbiol Biotechnol. 2009; 36(8): 1063-1072.
doi:10.1007/s10295-009-0590-3
Rathika R, Janaki V, Shanthi K, Kamala-Kannan S. Bioconversion of agro-industrial effluents for polyhydroxy-alkanoates production using Bacillus subtilis RS1. Int J Environ Sci Technol. 2019; 16(10): 5725-5734
doi:10.1007/s13762-018-2155-3
Mohapatra S, Sarkar B, Samantaray DP, Daware A, Maity S, Pattnaik S, Bhattacharjee S. Bioconversion of fish solid waste into PHB using Bacillus subtilis based submerged fermentation process. Environ Technol. 2017; 38(24): 3201-3208.
doi:10.1080/09593330.2017.1291759
Saranya Devi E, Vijayendra SVN, Shamala TR. Exploration of rice bran, an agro-industry residue, for the production of intra- and extra-cellular polymers by Sinorhizobium meliloti MTCC 100. Biocatal Agric Biotechnol. 2012; 1(1):80-84.
doi: 10.1016/j.bcab.2011.08.014
Narayanan A, Sajeev Kumar VA, Ramana KV. Production and characterization of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) from Bacillus mycoides DFC1 using rice husk hydrolyzate. Waste Biomass Valori. 2014; 5(1):109-118.
doi:10.1007/s12649-013-9213-3
Singh G, Kumari A, Mittal A, Yadav A, Aggarwal NK. Poly β-hydroxybutyrate production by Bacillus subtilis NG220 using sugar industry waste water. Biomed Res Int. 2013; 2013:1-10.
doi:10.1155/2013/952641
Haas R, Jin B, Zepf FT. Production of poly (3-hydro-xybutyrate) from waste potato starch. Biosci Biotechnol Biochem. 2008; 72 (1): 253-256.
doi:10.1271/bbb.70503
Verlinden RA, Hill DJ, Kenward MA, Williams CD, Piotrowska-Seget Z, Radecka IK. Production of polyhydroxyalkanoates from waste frying oil by Cupriavidus necator. AMB Express. 2011; 1(1): 11.
doi:10.1186/2191-0855-1-11
Prabu CS, Murugesan AG. Effective utilization and management of coir industrial waste for the production of poly- β- hydroxybutyrate (PHB) using the bacterium Azoto-bacter beijerinickii. Int J Environ Res 2010; 4(3): 519-524.
Hong K, Leung YC, Kwok SY, Law KH, Lo WH, Chua H, Yu PH. Construction of recombinant Escherichia coli strains for polyhydroxybutyrate production using soy waste as nutrient. Appl Biochem Biotechnol. 2000; 84-86:381-390.
doi:10.1385/abab:84-86:1-9:381
Sathish A, Glaittli K, Sims RC, Miller CD. Algae biomass based media for poly (3-hydroxybutyrate) (PHB) production by Escherichia coli. J Polym Environ. 2014; 22(2): 272-277.
doi:10.1007/s10924-014-0647-x
Al-Battashi H, Annamalai N, Al-Kindi S, Nair AS, Al-Bahry S, Verma JP, Sivakumar N. Production of bioplastic (poly-3-hydroxybutyrate) using waste paper as a feedstock: Optimization of enzymatic hydrolysis and fermentation employing Burkholderia sacchari. J Clean Prod. 2019; 214: 236-247.
doi: 10.1016/j.jclepro.2018.12.239
Wang B, Sharma-Shivappa RR, Olson JW, Khan SA. Production of polyhydroxybutyrate (PHB) by Alcaligenes latus using sugarbeet juice. Ind Crops Prod. 2013; 43:802-811.
doi: 10.1016/j.indcrop.2012.08.011
Umesh M, Priyanka K, Thazeem B, Preethi K. Production of single cell protein and polyhydroxyalkanoate from Carica papaya waste. Arab J Sci Eng. 2017; 42(6):2361-2369.
doi:10.1007/s13369-017-2519-x
Umesh M, Mani VM, Thazeem B, Preethi K. Statistical optimization of process parameters for bioplastic (PHA) production by Bacillus subtilis NCDC0671 using orange peel-based medium.Iran J Sci Technol Trans A Sci. 2018; 42(4): 1947-1955.
doi:10.1007/s40995-017-0457-9
Patel SKS, Kumar P, Singh M, Lee J-K, Kalia VC. Integrative approach to produce hydrogen and polyhydroxybutyrate from biowaste using defined bacterial cultures. Bioresour Technol. 2015; 176:136-141.
doi:10.1016/j.biortech.2014.11.029
Rebocho AT, Pereira JR, Freitas F, Neves LA, Alves VD, Sevrin C, Grandfils C, Reis MAM. Production of medium-chain length polyhydroxyalkanoates by Pseudomonas citronellolis grown in apple pulp waste. Appl Food Biotechnol. 2019; 6(1):71-82.
doi:10.22037/afb.v6i1.21793
Pernicova I, Enev V, Marova I, Obruca S. Interconnection of waste chicken feather biodegradation and keratinase and mcl-PHA production employing Pseudomonas putida KT2440. Appl Food Biotechnol. 2019; 6(1):83-90.
doi:10.22037/afb.v6i1.21429
Bustamante D, Tortajada M, Ramon D, Rojas A. Camelina oil as a promising substrate for mcl-PHA production in Pseudo-monas sp. Cultures. Appl Food Biotechnol. 2019; 6(1): 61-70.
doi:10.22037/afb.v6i1.21635
Kumar P, Kim BS. Paracoccus sp. Strain LL1 as a single cell factory for the conversion of waste cooking oil to polyhydroxyalkanoates and carotenoids. Appl Food Biotechnol. 2019; 6(1): 53-60.
doi:10.22037/afb.v6i1.21628
Tran NT, Li Z, Wang S, Zheng H, Aweya JJ, Wen X, Li S. Progress and perspectives of short‐chain fatty acids in aquaculture. Rev Aquacult. 2020; 12(1):283-298.
doi: 10.1111/raq.12317
Louis P, Hold GL, Flint HJ. The gut microbiota, bacterial metabolites and colorectal cancer. Nat Rev Microbiol. 2014; 12(10): 661-672.
doi:10.1038/nrmicro3344
Honda K, Littman DR. The microbiota in adaptive immune homeostasis and disease. Nature 2016; 535(7610):75-84.
doi:10.1038/nature18848
Najdegerami EH. Immunostimulatory and growth-promoting potential of poly-β-hydroxybutyrate in rainbow trout (Oncorhynchus mykiss) fingerlings culture. Iran J Fish Sci. 2020;19(2): 847-65
- Abstract Viewed: 1258 times
- pdf Downloaded: 832 times
