Bacterial Growth Enhancement Value and Nutritional Quality of Compounded Milk Media from Commercial Milk Brands
Applied Food Biotechnology,
Vol. 8 No. 1 (2021),
14 December 2020
,
Page 31-37
https://doi.org/10.22037/afb.v8i1.29607
Abstract
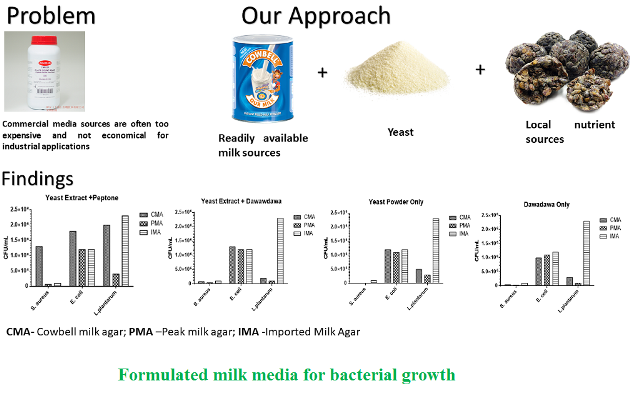
Background and Objective: Milk agar is recommended for enumeration of microorganisms in dairy products. Most of the commonly used media are imported to Nigeria and hence available in-house substitutes are necessary. In this study, microbial growth enhancement values of compounded milk agar were compared to imported milk agar using two milk brands. Dawadawa (fermented locust beans), yeast powder and peptone in the proportions of 1: 3: 5, respectively, were used as nutrient sources.
Material and Methods: Growth enhancement values of the compounded media were assessed on three bacterial species of Staphylococcus aureus, Escherichia coli and Lactobacillus plantarum. Furthermore, imported milk agar served as the control.
Results and Conclusion: Significant differences were seen in nutritional contents of the compounded milk agar and imported milk agar as well as the elemental compositions of compounded milk agar and imported milk agar. No significant differences were seen between the total viable counts of Staphylococcus aureus and Lactobacillus plantarum on compounded milk agar and imported milk agar. However, significant differences were reported in the total viable counts of Escherichia coli on compounded milk agar and imported milk agar. Media containing Cowbell milk powder recorded the highest total viable counts (1.3 ´ 105, 1.8 ´ 106 and 2.0 ´ 106 CFU ml-1) for Staphylococcus aureus, Escherichia coli and Lactobacillus plantarum, respectively. In conclusion, the Cowbell milk powder in combination with yeast extract and peptone is the best milk of choice for compounding milk agar when using local resources.
Conflict of interest: The authors declare no conflict of interest.
- ▪ Bacterial growth ▪ Compounded media ▪ Imported milk agar ▪ Milk brands
How to Cite
References
1. Jalili-Nik M, Shahri ZS, Hashemy SI, Mashkani B. Effects of major ingredients in cattle milk on enzyme kinetics of recombinant β-galactosidase (BGalP) expressed in Pichia pastoris. Appl Food Biotechnol. 2018; 5(4): 205-212.
doi:10.22037/afb.v5i4.22107.
Roberts TA, Cordier JL, Gram L, Tompkin RB, Pitt JI, Gorris LGM, Swanson KMJ. Milk and Dairy Products. In: Roberts TA, Cordier JL, Gram L, Tompkin RB, Pitt JI, Gorris LGM, Swanson KMJ (eds). Microorganisms in Foods 6. Springer, Boston, MA, 2005: pp. 643-715
Gorska-Warsewicz H, Rejman K, Laskowski W, Czeczotko M. Milk and dairy products and their nutritional contribution to the average polish diet. Nutrients 2019; 11(8):1771.
doi: 10.3390/nu11081771
Flint S, Bremer P, Brooks J, Palmer J, Sadiq FA, Seale B, Teh KH, Wu S, Md Zain SN. Bacterial fouling in dairy processing. Int Dairy J. 2020; 101: 104593.
doi: 10.1016/j.idairyj.2019.104593.
Pal M, Alemu J, Mulu S, Karanfil O, Parmar BC, Nayak JB. Microbial and Hygienic aspects of Dry Milk Powder. Beverage Food World. 2016; 43(7): 28-31.
Kivanc M, Evrim YE. Survival of Escherichia coli O157:H7 and Staphylococcus aureus during the fermentation and storage of kefir. Food Sci Technol. 2019; 39: 225-230.
doi: 10.1590/fst.39517.
Sadhu SP. Effect of cold chain interruptions on the shelf-life of fluid pasteurised skim milk at the consumer stage. Brazilian J Food Technol. 2018; 21: e2017064.
doi: 0.1590/1981-6723.06417.
Ryan K, Ray G. Medical Microbiology. 4th Edition, McGraw Hill York, 2004; pp. 120-123.
Esan EB, Muyiwa AA, Lawal JO. Performance of tea (Camellia sinensis L.) on culture media modified with locally sourced substitutes. J Anim Plant Sci. 2009; 4(1): 298-303.
AOAC. Official Methods of Analysis. 15th Edition. Association of Official Analytical Chemists, Washington DC, USA. 1990; 807-928.
Okafor N. Industrial Media and the Nutrition of Industrial Organisms In: Modern Microbiology and Biotechnology. Science Publishers, Enfield, New Hamsphire, USA. 2007; pp. 54-55.
Merchant SS, Helmann JD. Elemental economy: Microbial strategies for optimizing growth in the face of nutrient limitation. Adv Microb Physiol. 2012; 60: 91-210.
doi: 10.1016/B978-0-12-398264-3.00002-4
Basu S, Bose C, Ojha N, Das N, Das J, Pal M. Khurana S. Evolution of bacterial and fungal growth media. Bio-informatics. 2015; 11(4): 182-184.
doi: 10.6026/97320630011182
Yuan Y, Zhao B, Zhou S, Zhong S, Zhuang L. Electrocatalytic activity of anodic biofilm responses to pH changes in microbial fuel cells. Bioresour Technol. 2011; 102(13): 6887- 6891.
doi: 10.1016/j.biortech.2011.04.008.
Stainer RY, Ingraham JL, Wheelis ML, Painter PR. The Microbial World. 5th Edition, Prentice Hall Press, Englewood Cliffs, New Jersey, 1986; 689 pp.
McNamara PJ, Proctor RA. Staphylococcus aureus small colony variants, electron transport and persistent infections. Int J Antimicrob Agents, 2000; 2: 117-122.
doi: 10.1016/S0924-8579(99)00170-3.
Lodish H, Berk A, Zipursky SL, Matsudaira P, Baltimore D, Darnell J. Molecular Cell Biology. 4th Edition. Freeman WH; New York, 2000; 1084 pp. Available from: https://w-ww.ncbi.nlm.nih.gov/books/NBK21475/
Hebert EM, Raya RR, De Giori GS. Nutritional requirements of Lactobacillus delbrueckii subsp. lactis in a chemically defined medium. Curr Microbiol. 2004; 49(5): 341-345.
doi: 10.1007/s00284-004-4357-9
Tripp HJ, Kitner JB, Schwalbach MS, Dacey JW, Wilhelm LJ, Giovannoni SJ. SAR11 marine bacteria require exogenous reduced sulphur for growth. Nature 2008; 452: 741-744.
doi: 10.1038/nature06776
Toikkanen O, Outinen M, Malafronte L, Rojas OJ. Formation and structure of insoluble particles in reconstituted model infant formula powders. Int Dairy J. 2018; 82: 19-27.
doi: 10.1016/j.idairyj.2018.03.001
- Abstract Viewed: 605 times
- pdf Downloaded: 636 times
