Soluble Expression of Recombinant Human Bone Morphogenetic Protein-7 (rhBMP-7) in Escherichia coli Using SUMO Fusion System Soluble expression of rhBMP-7 in E. coli using SUMO fusion
Trends in Peptide and Protein Sciences,
Vol. 8 No. 1 (2023),
24 Dey 2023
,
Page 1-12 (e5)
https://doi.org/10.22037/tpps.v8i1.42962
Abstract
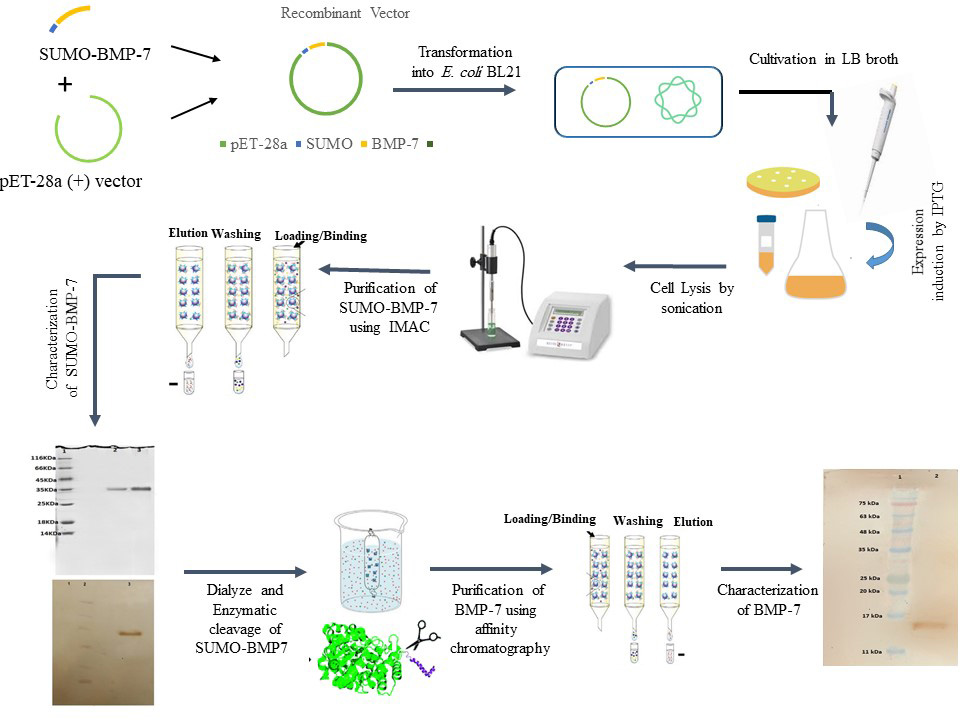
BMPs belong to transforming growth factor β superfamilies, which their principal role is inducing bone and cartilage formation at heterotopic and orthotopic sites. Since the formation of inclusion bodies is the main limitation of producing these proteins in Escherichia coli, in this study, the small ubiquitin-like modifiers (SUMO) fusion system was employed to improve solubility and expression of recombinant human BMP-7 (rhBMP-7) in E. coli. The SUMO fusion system has the ability to enhance protein expression, reduce target protein proteolytic degradation, and increase protein folding and solubility. In the current study, the SUMO protein gene was fused to the N-terminus of the BMP-7 gene, and cloned in the pET-28a vector. After purification of the expressed SUMO-BMP-7 protein by Ni-NTA chromatography, SUMO was removed from the BMP-7 protein using SUMO protease. In the second step of purification using Ni-NTA chromatography, the cleaved BMP-7 protein was purified and then identified by Western blot analysis. The results of the current study demonstrated that the SUMO fusion system is able to increase the soluble form of rhBMP-7. Furthermore, rhBMP-7 can be purified by a two-step purification strategy including: 1) purification of SUMO-BMP-7 and 2) purification of rBMP-7 after cleavage using Ni-NTA chromatography. Altogether, this research has provided a feasible approach for large-scale production of soluble rhBMP-7, to facilitate its further medical development.
HIGHLIGHTS
- SUMO is a well characterized family of ubiquitin-like molecules.
- SUMO fusion led to increased expression and solubility of BMP-7.
- BMP-7 is involved in the process of bone formation.
- BMP-7
- Escherichia coli
- Recombinant protein
- Soluble expression
- SUMO fusion system
How to Cite
References
Acuña, M. L. (2022). ″The Contribution of Alternative Splicing and Post-Transcriptional Mechanisms Toward the Global Control of SUMO1/2/3 SUMOylation.″ Doctoral Dissertation, The University of Texas at El Paso.
Aslani, S., Abhari, A., Sakhinia, E., Sanajou, D., Rajabi, H. and S. Rahimzadeh, (2019). "Interplay between microRNAs and Wnt, transforming growth factor‐β, and bone morphogenic protein signaling pathways promote osteoblastic differentiation of mesenchymal stem cells." Journal of Cellular Physiology, 234(6): 8082-8093. DOI: https://doi.org/10.1002/jcp.27582.
Begam, H., Nandi, S. K., Kundu, B. and A. Chanda, (2017). "Strategies for delivering bone morphogenetic protein for bone healing." Materials Science and Engineering: C, 70(1): 856-869. DOI: https://doi.org/10.1016/j.msec.2016.09.074.
Bessa, P. C., Cerqueira, M., Rada, T., Gomes, M. E., Neves, N., Nobre, A., Reis, R. and M. Casal, (2009). "Expression, purification and osteogenic bioactivity of recombinant human BMP-4,-9,-10,-11 and-14." Protein Expression and Purification, 63(2): 89-94. DOI: https://doi.org/10.1016/j.pep.2008.09.014.
Bhatwa, A., Wang, W., Hassan, Y. I., Abraham, N., Li, X. Z. and T. Zhou, (2021). "Challenges associated with the formation of recombinant protein inclusion bodies in Escherichia coli and strategies to address them for industrial applications." Frontiers in Bioengineering and Biotechnology, 9: 630551. DOI: https://doi.org/10.3389/fbioe.2021.630551.
Blokhuis, T. J., Calori, G. M. and G. Schmidmaier, (2013). "Autograft versus BMPs for the treatment of non-unions: what is the evidence?" Injury, 44: S40-S42. DOI: https://doi.org/10.1016/S0020-1383(13)70009-3.
Ceylan, H. and O. Erdoğan, (2017). "Cloning, expression, and characterization of human brain acetylcholinesterase in Escherichia coli using a SUMO fusion tag." Turkish Journal of Biology, 41(1): 77-87. DOI: https://doi.org/ 10.3906/biy-1602-83.
Chen, B. L., Peng, J., Li, Q. F., Yang, M., Wang, Y. and W. Chen, (2013). "Exogenous bone morphogenetic protein-7 reduces hepatic fibrosis in Schistosoma japonicum-infected mice via transforming growth factor-β/Smad signaling." World Journal of Gastroenterology, 19(9): 1405-1415. DOI: https://doi.org/10.3748/wjg.v19.i9.1405.
Chen, Q. C., Liu, L., Yu, T. Y., Tang, L., Yin, M. L., Zhu, W. H., Jiang, X. Y. and H. Y. Wang, (2021). "High-level expression and purification of melittin in Escherichia coli using SUMO fusion partner." International Journal of Peptide Research and Therapeutics, 27: 9-15. DOI: https://doi.org/10.1007/s10989-020-10060-4.
Chen, X., Xu, J., Jiang, B. and D. Liu, (2016). "Bone morphogenetic protein-7 antagonizes myocardial fibrosis induced by atrial fibrillation by restraining transforming growth factor-β (TGF-β)/Smads signaling." Medical Science Monitor, 22: 3457-3468. DOI: https://doi.org/10.12659/msm.897560.
Cheng, C., Wu, S., Cui, L., Wu, Y., Jiang, T. and B. He, (2017). "A novel Ffu fusion system for secretory expression of heterologous proteins in Escherichia coli." Microbial Cell Factories, 16(1): 231. DOI: https://doi.org/10.1186/s12934-017-0845-z.
de Marco, A. (2020). "Recombinant expression of nanobodies and nanobody-derived immunoreagents." Protein Expression and Purification, 172: 105645. DOI: https://doi.org/10.1016/j.pep.2020.105645.
Deng, Z., Li, Y., Gao X., Lei, G. and J. Huard, (2018). "Bone morphogenetic proteins for articular cartilage regeneration." Osteoarthritis and Cartilage, 26(9): 1153-1161. DOI: https://doi.org/10.1016/j.joca.2018.03.007.
Dugmehchi, A., Sadipour, G., Talebkhan, Y., Jahandar, H., Nemati, F., Mohit, E. and L. Nematollahi, (2022). "Cytoplasmic expression of human bone morphogenetic protein-7 by a genetically engineered strain of Escherichia coli, SHuffle® Strain." Trends in Peptide and Protein Sciences, 7: 1-7 (e7). DOI: https://doi.org/10.22037/tpps.v7i.39039.
Epstein, N. E. (2013). "Commentary on research of bone morphogenetic protein discussed in review article: Genetic advances in the regeneration of the intervertebral disc." Surgical Neurology International, 4(Suppl 2): S106-8. DOI: https://doi.org/ 10.4103/2152-7806.109452.
Fazen, C. H., Kahkoska, A. R. and R. P. Doyle, (2012). "Expression and purification of human PYY (3–36) in Escherichia coli using a His-tagged small ubiquitin-like modifier fusion." Protein Expression and Purification, 85(1): 51-59. DOI: https://doi.org/10.1016/j.pep.2012.06.015.
Gazzerro, E. and C. Minetti, (2007). "Potential drug targets within bone morphogenetic protein signaling pathways." Current Opinion in Pharmacology, 7(3): 325-333. DOI: https://doi.org/10.1016/j.coph.2007.01.003.
Gillman, C. E. and A. C. Jayasuriya, (2021). "FDA-approved bone grafts and bone graft substitute devices in bone regeneration." Materials Science and Engineering: C, 130: 112466. DOI: https://doi.org/10.1016/j.msec.2021.112466.
Guerrero, F., Ciragan, A. and H. Iwaï, (2015). "Tandem SUMO fusion vectors for improving soluble protein expression and purification." Protein Expression and Purification, 116: 42-49. DOI: https://doi.org/ 10.1016/j.pep.2015.08.019.
Gupta, S. K. and P. Shukla, (2016). "Advanced technologies for improved expression of recombinant proteins in bacteria: perspectives and applications." Critical Reviews in Biotechnology, 36(6): 1089-1098. DOI: https://doi.org/10.3109/07388551.2015.1084264.
Hajihassan, Z., Sohrabi, M., Rajabi Bazl, M. and H. Eftekhary, (2016). "Expression of human nerve growth factor beta and bacterial protein disulfide isomerase (DsbA) as a fusion protein (DsbA:: hNGF) significantly enhances periplasmic production of hNGF beta in Escherichia coli." Romanian Biotechnological Letters, 21(5): 11850-11856.
Hanif, M. U., Yaseen, A., Gul, R., Mirza, M. U., Nawaz, M. H., Ahmed, S. S., Aziz, S., Chaudhary, S., Khan, A. A. and M. Shoaib, (2018). "Small ubiquitin-like modifier protein 3 enhances the solubilization of human bone morphogenetic protein 2 in E. coli." Applied Biochemistry and Biotechnology, 186: 256-270. DOI: https://doi.org/10.1007/s12010-018-2736-0.
Hasan, T. and D. Saluja, (2015). "Structural allostery and protein–protein interactions of Sin3." Proteostasis and Chaperone Surveillance, 3-24. DOI: https://doi.org/10.1007/978-81-322-2467-9_1.
Haversath, M., Catelas, I., Li, X., Tassemeier, T. and M. Jäger, (2012). "PGE2 and BMP-2 in bone and cartilage metabolism: 2 intertwining pathways." Canadian Journal of Physiology and Pharmacology, 90(11): 1434-1445. DOI: https://doi.org/ 10.1139/y2012-123.
Hilgarth, R. S. and K. D. Sarge, (2005). "Detection of sumoylated proteins." Ubiquitin-Proteasome Protocols, 329-337. DOI: https://doi.org/10.1385/1-59259-895-1:329.
Hou, H., Yan, W., Du, K., Ye, Y., Cao, Q. and W. Ren, (2013). "Construction and expression of an antimicrobial peptide scolopin 1 from the centipede venoms of Scolopendra subspinipes mutilans in Escherichia coli using SUMO fusion partner." Protein Expression and Purification, 92(2): 230-234. DOI: https://doi.irg/10.1016/j.pep.2013.10.004.
Jeong, T. H., Son, Y. J., Ryu, H. B., Koo, B. K., Jeong, S. M., Hoang, P., Do, B. H., Song, J. A., Chong, S. H. and R. C. Robinson, (2014). "Soluble expression and partial purification of recombinant human erythropoietin from E. coli." Protein Expression and Purification, 95: 211-218. DOI: https://doi.org/10.1016/j.pep.2014.01.001.
Karyolaimos, A., K. Dolata, M., Antelo-Varela, M., Mestre Borras, A., Elfageih, R., Sievers, S., Becher, D., Riedel, K. and J. W. de Gier, (2020). "Escherichia coli can adapt its protein translocation machinery for enhanced periplasmic recombinant protein production." Frontiers in Bioengineering and Biotechnology, 7: 465. DOI: https://doi.org/10.3389/fbioe.2019.00465.
Khorsand, B., Elangovan, S., Hong, L., Dewerth, A., Kormann, M. S. and A. K. Salem, (2017). "A comparative study of the bone regenerative effect of chemically modified RNA encoding BMP-2 or BMP-9." The AAPS Journal, 19: 438-446. DOI: https://doi.org/10.1208/s12248-016-0034-8.
Ki, M. R. and S. P. Pack, (2020). "Fusion tags to enhance heterologous protein expression." Applied Microbiology and Biotechnology, 104(6): 2411-2425. DOI: https://doi.org/10.1007/s00253-020-10402-8.
Ko, H., Kang, M., Kim, M. J., Yi, J., Kang, J., Bae, J. H., Sohn, J. H. and B. H. Sung, (2021). "A novel protein fusion partner, carbohydrate-binding module family 66, to enhance heterologous protein expression in Escherichia coli." Microbial Cell Factories, 20: 1-12. DOI: https://doi.org/10.1186/s12934-021-01725-w.
Kübler, N., Würzler, K., Reuther, J., Sieber, E., Kirchner, T. and W. Sebald, (2000). "Effect of different factors on the bone forming properties of recombinant BMPs." Mund-, Kiefer-und Gesichtschirurgie: MKG, 4: S465-469. DOI: https://doi.org/ 10.1007/PL00012693.
Kuo, D., Nie, M., De Hoff, P., Chambers, M., Phillips, M., Hirsch, A. M. and A. J. Courey, (2011). "A SUMO–Groucho Q domain fusion protein: Characterization and in vivo Ulp1-mediated cleavage." Protein Expression and Purification, 76(1): 65-71. DOI: https://doi.org/10.1016/j.pep.2010.08.008.
Lee, C. D., Yan, Y. P., Liang, S. M. and T. F. Wang, (2009). "Production of FMDV virus-like particles by a SUMO fusion protein approach in Escherichia coli." Journal of Biomedical Science, 16(1): 1-7. DOI: https://doi.org/10.1186/1423-0127-16-69.
Lee, S. (2022). "Peptide ligand interaction with maltose-binding protein tagged to the calcitonin gene-related peptide receptor: The inhibitory role of receptor N-glycosylation." Peptides, 150: 170735. DOI: https://doi.org/10.1016/j.peptides.2022.170735.
Li, J., Han, Q., Zhang, T., Du, J., Sun, Q. and Y. Pang, (2018). "Expression of soluble native protein in Escherichia coli using a cold-shock SUMO tag-fused expression vector." Biotechnology Reports, 19: e00261. DOI: https://doi.org/10.1016/j.btre.2018.e00261.
Liew, O. W., Ang, C. X., Peh, Y. P., Chong, P. C. J., Ng, Y. X., Hwang, L. A., Koh, X. Y., Yip, Y. M., Liu, W. and A. M. Richards, (2014). "A His6-SUMO-eXact tag for producing human prepro-urocortin 2 in Escherichia coli for raising monoclonal antibodies." Journal of Immunological Methods, 403(1-2): 37-51. DOI: https://doi.org/10.1016/j.jim.2013.11.015.
Linova, M. Y., Risør, M. W., Jørgensen, S. E., Mansour, Z., Kaya, J., Sigurdarson, J. J., Enghild, J. J. and H. Karring, (2020). "A novel approach for production of an active N-terminally truncated Ulp1 (SUMO protease 1) catalytic domain from Escherichia coli inclusion bodies." Protein Expression and Purification, 166: 105507. DOI: https://doi.org/10.1016/j.pep.2019.105507.
Liu, Y., Zhang, W., Yang, X., Kang, G., Wang, D. and H. Huang, (2015). "DsbA-DsbA mut fusion chaperon improved soluble expression of human trypsinogen-1 in Escherichia coli." Frontiers of Chemical Science and Engineering, 9: 511-521. DOI: https://doi.org/10.1007/s11705-015-1519-1.
Malik, A. (2016). "Protein fusion tags for efficient expression and purification of recombinant proteins in the periplasmic space of E. coli." 3 Biotech, 6(1): 44. DOI: https://doi.org/10.1007/s13205-016-0397-7.
Mo, Q., Fu, A., Lin, Z., Wang, W., Gong, L. and W. Li, (2018). "Expression and purification of antimicrobial peptide AP2 using SUMO fusion partner technology in Escherichia coli." Letters in Applied Microbiology, 67(6): 606-613. DOI: https://doi.org/10.1111/lam.13079.
Moghaddam, A., Elleser, C., Biglari, B., Wentzensen, A. and G. Zimmermann, (2010). "Clinical application of BMP 7 in long bone non-unions." Archives of Orthopaedic and Trauma Surgery, 130(1): 71-76. DOI: https://doi.org/ 10.1007/s00402-009-0982-x.
Namvar, S., Barkhordari, F., Raigani, M., Jahandar, H., Nematollahi, L. and F. Davami, (2018). "Cloning and soluble expression of mature α-luffin from Luffa cylindrica in E. coli using SUMO fusion protein." Turkish Journal of Biology, 42(1): 23-32. DOI: https://doi.org/10.3906/biy-1708-12.
Nasrabadi, D., Rezaeiani, S., Sayadmanesh, A., Eslaminejad, M. B. and A. Shabani, (2018). "Inclusion body expression and refolding of recombinant bone morphogenetic protein-2." Avicenna Journal of Medical Biotechnology, 10(4): 202-207. PMCID: PMC6252035.
Neuerburg, C., M. Mittlmeier, L., Keppler, A. M., Westphal, I., Glass, Ä., Saller, M. M., Herlyn, P. K., Richter, H., Böcker, W. and M. Schieker, (2019). "Growth factor-mediated augmentation of long bones: evaluation of a BMP-7 loaded thermoresponsive hydrogel in a murine femoral intramedullary injection model." Journal of Orthopaedic Surgery and Research, 14(1): 1-10. DOI: https://doi.org/10.1186/s13018-019-1315-6.
Peciak, K., Tommasi, R., Choi, J. w., Brocchini, S. and E. Laurine, (2014). "Expression of soluble and active interferon consensus in SUMO fusion expression system in E. coli." Protein Expression and Purification, 99: 18-26. DOI: https://doi.org/10.1016/j.pep.2014.03.009.
Pratheesh, P. T., Nimisha, S., Jess, V., Asha, K. and R. K. Agarwal, (2019). "Expression and purification of an immunogenic SUMO-OmpC fusion protein of Salmonella Typhimurium in Escherichia coli." Biologicals, 62: 22-26. DOI: https://doi.org/10.1016/j.biologicals.2019.10.010.
Rao, S. M., Ugale, G. M. and S. B. Warad, (2013). "Bone morphogenetic proteins: periodontal regeneration." North American Journal of Medical Sciences, 5(3): 161. DOI: https://doi.org/10.4103/1947-2714.109175.
Raran‐Kurussi, S. and D. S. Waugh, (2014). "Unrelated solubility‐enhancing fusion partners MBP and NusA utilize a similar mode of action." Biotechnology and Bioengineering, 111(12): 2407-2411. DOI: https://doi.org/10.1002/bit.25317.
Rezaie, F., Davami, F., Mansouri, K., Agha Amiri, S., Fazel, R., Mahdian, R., Davoudi, N., Enayati, S., Azizi, M. and V. Khalaj, (2017). "Cytosolic expression of functional Fab fragments in Escherichia coli using a novel combination of dual SUMO expression cassette and EnBase® cultivation mode." Journal of Applied Microbiology, 123(1): 134-144. DOI: https://doi.org/10.1111/jam.13483.
Sadr, V., Saffar, B. and R. Emamzadeh, (2017). "Functional expression and purification of recombinant Hepcidin25 production in Escherichia coli using SUMO fusion technology." Gene, 610: 112-117. DOI: https://doi.org/10.1016/j.gene.2017.02.010.
Saffari, B., Amininasab, M., Sheikhi, S. and J. Davoodi, (2020). "An efficient method for recombinant production of human alpha synuclein in Escherichia coli using thioredoxin as a fusion partner." Preparative Biochemistry & Biotechnology, 50(7): 723-734. DOI: https://doi.org/10.1080/10826068.2020.1734938.
Sampath, T. K. and A. H. Reddi, (2020). "Discovery of bone morphogenetic proteins–A historical perspective." Bone, 140: 115548. DOI: https://doi.org/10.1016/j.bone.2020.115548.
Sandler, A. B., Scanaliato, J. P., Raiciulescu, S., Nesti, L. and J. C. Dunn, (2023). "Bone morphogenic protein for upper extremity fractures: a systematic review." Hand, 18(1): 80-88. DOI: https://doi.org/ 10.1177/1558944721990805.
Sang, M., Xu, C., Wei, Z., Wu, X., Guo, Y., Li, J., Wang, Z. and J. Zhang, (2019). "Cloning and high-level SUMO-mediated fusion expression of a serine protease inhibitor from Hyphantria cunea (Drury) that exhibits activity against papain." Protein Expression and Purification, 158: 36-43. DOI: https://doi.org/10.1016/j.pep.2019.02.011.
Satakarni, M. and R. Curtis, (2011). "Production of recombinant peptides as fusions with SUMO." Protein Expression and Purification, 78(2): 113-119. DOI: https://doi.org/10.1016/j.pep.2011.04.015.
Sheikh, Z., Javaid, M. A., Hamdan, N. and R. Hashmi, (2015). "Bone regeneration using bone morphogenetic proteins and various biomaterial carriers." Materials, 8(4): 1778-1816. DOI: https://doi.org/ 10.3390/ma8041778.
Sriramachandran, A. M. and R. J. Dohmen, (2014). "SUMO-targeted ubiquitin ligases." Biochimica et Biophysica Acta (BBA)-Molecular Cell Research, 1843(1): 75-85. DOI: https://doi.org/10.1016/j.bbamcr.2013.08.022.
Sun, W., Xie, J., Lin, H., Mi, S., Li, Z., Hua, F. and Z. Hu, (2012). "A combined strategy improves the solubility of aggregation-prone single-chain variable fragment antibodies." Protein Expression and Purification, 83(1): 21-29. DOI: https://doi.org/10.1016/j.pep.2012.02.006.
Sun, Z., Xia, Z., Bi, F. and J. N. Liu, (2008). "Expression and purification of human urodilatin by small ubiquitin-related modifier fusion in Escherichia coli." Applied Microbiology and Biotechnology, 78: 495-502. DOI: https://doi.org/10.1007/s00253-007-1330-0.
Tileva, M., Krachmarova, E., Ivanov, I., Maskos, K. and G. Nacheva, (2016). "Production of aggregation prone human interferon gamma and its mutant in highly soluble and biologically active form by SUMO fusion technology." Protein Expression and Purification, 117: 26-34. DOI: https://doi.org/10.1016/j.pep.2015.09.022.
Truong, L., Hevener, K. E., Rice, A. J., Patel, K., Johnson, M. E. and H. Lee, (2013). "High-level expression, purification, and characterization of Staphylococcus aureus dihydroorotase (PyrC) as a cleavable His-SUMO fusion." Protein Expression and Purification, 88(1): 98-106. DOI: https://doi.org/10.1016/j.pep.2012.11.018.
Upadhyay, S. K., Saurabh, S., Rai, P., Singh, R., Chandrashekar, K., Verma, P. C., Singh, P. and R. Tuli, (2010). "SUMO fusion facilitates expression and purification of garlic leaf lectin but modifies some of its properties." Journal of Biotechnology, 146(1-2): 1-8. DOI: https://doi.org/10.1016/j.jbiotec.2010.01.013.
Vu, T. T. T., Jeong, B., Yu, J., Koo, B. K., Jo, S. H., Robinson, R. C. and H. Choe, (2014). "Soluble prokaryotic expression and purification of crotamine using an N-terminal maltose-binding protein tag." Toxicon, 92: 157-165. DOI: https://doi.org/10.1016/j.toxicon.2014.10.017.
Wang, G., Han, J., Wang, S. and P. Li, (2014). "Expression and purification of recombinant human bone morphogenetic protein-7 in Escherichia coli." Preparative Biochemistry and Biotechnology, 44(1): 16-25. DOI: https://doi.org/ 10.1080/10826068.2013.782043.
Wang, Q., Min, C., Yan, T., Pu, H., Xin, Y., Zhang, S., Luo, L. and Z. Yin, (2011). "Production of glutamine synthetase in Escherichia coli using SUMO fusion partner and application to l-glutamine synthesis." World Journal of Microbiology and Biotechnology, 27: 2603-2610. DOI: https://doi.org/10.1007/s11274-011-0733-3.
Wang, Q., Zhao, J., Wang, Y., Sun, H., Jiang, Y., Luo, L. and Z. Yin, (2013). "Functional expression of hepassocin in Escherichia coli using SUMO fusion partner and molecular chaperones." Protein Expression and Purification, 92(2): 135-140. DOI: https://doi.org/10.1016/j.pep.2013.09.014.
Wang, Z., Li, H., Guan, W., Ling, H., Wang, Z., Mu, T., Shuler, F. D. and X. Fang, (2010). "Human SUMO fusion systems enhance protein expression and solubility." Protein Expression and Purification, 73(2): 203-208. DOI: https://doi.org/10.1016/j.pep.2010.05.001.
Wang, Z., Li, N., Wang, Y., Wu, Y., Mu, T., Zheng, Y., Huang, L. and X. Fang, (2012). "Ubiquitin-intein and SUMO2-intein fusion systems for enhanced protein production and purification." Protein Expression and Purification, 82(1): 174-178. DOI: https://doi.org/10.1016/j.pep.2011.11.017Get rights and content.
Williams, R. M., Harvey, J. D., Budhathoki-Uprety, J. and D. A. Heller, (2020). "Glutathione-S-transferase fusion protein nanosensor." Nano Letters, 20(10): 7287-7295. DOI: https://doi.org/10.1021/acs.nanolett.0c02691.
Xiao, W., Jiang, L., Wang, W., Wang, R. and J. Fan, (2018). "Evaluation of rice tetraticopeptide domain-containing thioredoxin as a novel solubility-enhancing fusion tag in Escherichia coli." Journal of Bioscience and Bioengineering, 125(2): 160-167. DOI: https://doi.org/10.1016/j.jbiosc.2017.08.016.
Yadav, D. K., Yadav, N., Yadav, S., Haque, S. and N. Tuteja, (2016). "An insight into fusion technology aiding efficient recombinant protein production for functional proteomics." Archives of Biochemistry and Biophysics, 612: 57-77. DOI: http://doi.org/10.1016/j.abb.2016.10.012.
Yang, B. C., Zhang, C., Wang, C., Zhou, H., Li, Z. Y., Song, Y. J., Zhang, T. C. and X. G. Luo, (2017). "Soluble expression and purification of heparinase I in Escherichia coli using a hexahistidine-tagged small ubiquitin-like modifier as a fusion partner." Biotechnology & Biotechnological Equipment, 31(5): 1040-1045. DOI: https://doi.org/10.1080/13102818.2017.1355264.
Ye, L. and W. G. Jiang, (2016). "Bone morphogenetic proteins in tumour associated angiogenesis and implication in cancer therapies." Cancer Letters, 380(2): 586-597. DOI: https://doi.org/10.1016/j.canlet.2015.10.036.
Ye, T., Lin, Z. and H. Lei, (2008). "High-level expression and characterization of an anti-VEGF165 single-chain variable fragment (scFv) by small ubiquitin-related modifier fusion in Escherichia coli." Applied Microbiology and Biotechnology, 81(2): 311-317. DOI: https://doi.org/10.1007/s00253-008-1655-3.
Yu, Z., Wang, Q., Ma, Q. and R. Zhang, (2013). "Secretory expression of lacticin Q fused with SUMO in Bacillus subtilis." Protein Expression and Purification, 89(1): 51-55. DOI: https://doi.org/10.1016/j.pep.2013.02.014.
Zang, S., Mu, R., Chen, F., Wei, X., Zhu, L., Han, B., Yu, H., Bi, B., Chen, B. and Q. Wang, (2019). "Injectable chitosan/β-glycerophosphate hydrogels with sustained release of BMP-7 and ornidazole in periodontal wound healing of class III furcation defects." Materials Science and Engineering: C, 99: 919-928. DOI: https://doi.org/10.1016/j.msec.2019.02.024.
Zhang, H., Wu, J., Zhang, Y., Fu, N., Wang, J. and S. Zhao, (2010). "Optimized procedure for expression and renaturation of recombinant human bone morphogenetic protein-2 at high protein concentrations." Molecular Biology Reports, 37(7): 3089-3095. DOI: https://doi.org/10.1007/s11033-009-9883-x.
Zhang, J., Ma, L. and S. Q. Zhang, (2014). "Expression and purification of soluble human APRIL in Escherichia coli using ELP-SUMO tag." Protein Expression and Purification, 95: 177-181. DOI: https://doi.org/10.1016/j.pep.2013.12.013.
Zia, M. A., Shah, M. S., Khan, R. S. A., Farooq, U., Shafi, J. and M. Habib, (2022). "High level expression and purification of recombinant 3ABC non-structural protein of foot-and-mouth disease virus using SUMO fusion system." Protein Expression and Purification, 191: 106025. DOI: https://doi.org/ 10.1016/j.pep.2021.106025.
- Abstract Viewed: 425 times
- PDF Downloaded: 73 times