Photoautotrophic Cultivation of Arthrospira maxima for Protein Accumulation under Minimum Nutrient Availability
Applied Food Biotechnology,
Vol. 7 No. 4 (2020),
18 August 2020
,
Page 225-234
https://doi.org/10.22037/afb.v7i4.30353
Abstract
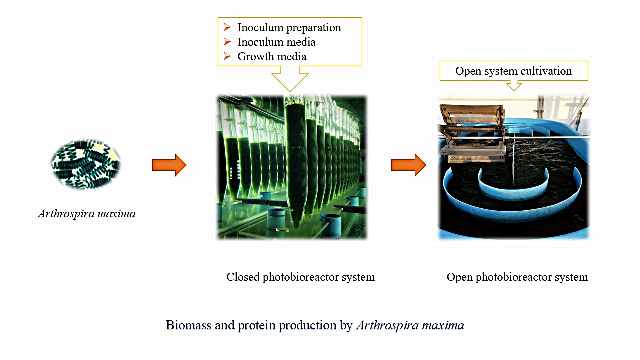
Background and Objective: Arthrospira cyanobacteria are important freshwater blue-green microalgae as food protein sources for humans. Arthrospira maxima phototrophic cultivation was carried out under various minimum media formulations to enhance biomass and protein productions. Macronutrients and micronutrients are vital for biomass production and protein accumulation of Arthrospira maxima for food supplemented uses.
Materials and Methods: Photoautotrophic cultivation of Arthrospira maxima IFRPD 1183 was carried out using various Zarrouk culture media and inoculum preparation conditions for biomass and protein productions. Arthrospira maxima IFRPD 1183 was cultivated using algal chamber under closed and open photobioreactor systems.
Results and Conclusion: Micronutrients of B6 solution; NH4VO3, K3Cr2 (SO4)4. 24H2O, NiSO4. 7H2O, Na2WO3, Co (NO3)2. 6H2O and Ti2 (SO4)3 did not affect cell growth and protein accumul-ation in absence of media for inoculum production. Inoculum preparation under various conditions of Arthrospira maxima IFRPD 1183 was studied using filtration of the old media before use (cell filtration inoculum) with no filtrations of the old media before use (cell non-filtration inoculum). Cell non-filtration inoculum preparation was reported appropriate for biomass production and protein accumulation. For large-scale productions, open pond system of Arthrospira maxima IFRPD 1183 resulted in maximum biomass and protein production at nearly 1 g l-1 and 64% (DW), respectively, for repeat batch photoautotrophic cultivation. Absence of micronutrients with cell non-filtration inoculum was reported as an easy process to achieve large biomass and protein productions for closed and open photoautotrophic cultivations of Arthrospira maxima at decreased costs.
- Arthrospira
- biomass
- Cyanobacteria
- Nutrients
- Photoautotrophic
- Protein
How to Cite
References
Pelizer LH, de Carvalho JCM, de Oliveira Moraes I. Protein production by Arthrospira (Spirulina) platensis in solid state cultivation using sugarcane bagasse as support. Biotechnol Reports [Internet]. 2015 Mar 1 [cited 2020 Feb 29];5:70–6. Available from: https://www.sciencedirect.com/science/article/pii/S2215017X14000629#bib0045
Nuhu AA. Spirulina (Arthrospira): An Important Source of Nutritional and Medicinal Compounds. J Mar Biol [Internet]. 2013;2013:8. Available from: http://dx.doi.org/10.1155/2013/325636
Borowitzka MA. Microalgae in Medicine and Human Health: A Historical Perspective. Microalgae Heal Dis Prev [Internet]. 2018 Jan 1 [cited 2020 Mar 1];195–210. Available from: https://www.sciencedirect.com/science/article/pii/B9780128114056000098
Karkos PD, Leong SC, Karkos CD, Sivaji N, Assimakopoulos DA. Spirulina in clinical practice: Evidence-based human applications. Evidence-based Complementary and Alternative Medicine. 2011.
Ku CS, Yang Y, Park Y, Lee J. Health benefits of blue-green algae: Prevention of cardiovascular disease and nonalcoholic fatty liver disease. Journal of Medicinal Food. 2013.
Finamore A, Palmery M, Bensehaila S, Peluso I. Antioxidant, Immunomodulating, and Microbial-Modulating Activities of the Sustainable and Ecofriendly Spirulina. Oxidative Medicine and Cellular Longevity. 2017.
Matos ÂP. Microalgae as a Potential Source of Proteins. Proteins Sustain Source, Process Appl [Internet]. 2019 Jan 1 [cited 2020 Mar 2];63–96. Available from: https://www.sciencedirect.com/science/article/pii/B9780128166956000039
Borowitzka MA. Biology of Microalgae. Microalgae Heal Dis Prev [Internet]. 2018 Jan 1 [cited 2020 Mar 2];23–72. Available from: https://www.sciencedirect.com/science/article/pii/B9780128114056000037
Chojnacka K, Noworyta A. Evaluation of Spirulina sp. growth in photoautotrophic, heterotrophic and mixotrophic cultures. Enzyme Microb Technol. 2004;
Aishvarya V, Pradhan N, Nayak RR, Sukla LB, Mishra BK. Enhanced inorganic carbon uptake by Chlorella sp. IMMTCC-2 under autotrophic conditions for lipid production and CO2 sequestration. J Appl Phycol [Internet]. 2012;24(6):1455–63. Available from: https://doi.org/10.1007/s10811-012-9801-9
Sun Z, Liu J, Zhou Z-G. Algae for biofuels. In: Handbook of Biofuels Production. 2016.
Costa JAV, de Morais MG. An Open Pond System for Microalgal Cultivation. In: Biofuels from Algae. 2013.
Furmaniak MA, Misztak AE, Franczuk MD, Wilmotte A, Waleron M, Waleron KF. Edible cyanobacterial genus Arthrospira: Actual state of the art in cultivation methods, genetics, and application in medicine. Front Microbiol. 2017;8(DEC):1–21.
Masojídek J, Torzillo G. Mass Cultivation of Freshwater Microalgae☆. In: Reference Module in Earth Systems and Environmental Sciences. 2014.
Delrue F, Alaux E, Moudjaoui L, Gaignard C, Fleury G, Perilhou A, et al. Optimization of Arthrospira platensis (Spirulina) growth: From laboratory scale to pilot scale. Fermentation. 2017;
Çelekli A, Yavuzatmaca M. Predictive modeling of biomass production by Spirulina platensis as function of nitrate and NaCl concentrations. Bioresour Technol. 2009;
Yuan X, Kumar A, Sahu AK, Ergas SJ. Impact of ammonia concentration on Spirulina platensis growth in an airlift photobioreactor. Bioresour Technol. 2011;
Sassano CEN, Gioielli LA, Ferreira LS, Rodrigues MS, Sato S, Converti A, et al. Evaluation of the composition of continuously-cultivated Arthrospira (Spirulina) platensis using ammonium chloride as nitrogen source. Biomass and Bioenergy. 2010;
Madkour FF, Kamil AEW, Nasr HS. Production and nutritive value of Spirulina platensis in reduced cost media. Egypt J Aquat Res. 2012;
Affan MA, Lee DW, Al-Harbi SM, Kim HJ, Abdulwassi NI, Heo SJ, et al. Variation of Spirulina maxima biomass production in different depths of urea-used culture medium. Brazilian J Microbiol. 2015;
Soni RA, Sudhakar K, Rana RS. Comparative study on the growth performance of Spirulina platensis on modifying culture media. Energy Reports. 2019;
Zarrouk C. Contribution à l’étude d’une Cyanophycée. Influence de Divers Facteurs Physiques et Chimiques sur la Croissanceet la Photosynthèse de Spirulina maxima. Ph. D. Thesis. University of Paris, France. 1996.
AOAC. Official Methods of Analysis of AOAC International. Association of Official Analysis Chemists International. 2005.
Yang J, Rasa E, Tantayotai P, Scow KM, Yuan H, Hristova KR. Mathematical model of Chlorella minutissima UTEX2341 growth and lipid production under photoheterotrophic fermentation conditions. Bioresour Technol [Internet]. 2011 Feb 1 [cited 2020 Feb 28];102(3):3077–82. Available from: https://www.sciencedirect.com/science/article/pii/S0960852410017050?via%3Dihub
Mosulishvili LM, Kirkesali EI, Belokobylsky AI, Khizanishvili AI, Frontasyeva M V., Pavlov SS, et al. Experimental substantiation of the possibility of developing selenium- and iodine-containing pharmaceuticals based on blue-green algae Spirulina platensis. J Pharm Biomed Anal. 2002;
Lu J, Yoshizaki G, Sakai K, Takeuchi T. Acceptability of raw Spirulina platensis by larval tilapia Oreochromis niloticus. Fish Sci. 2002;
Pagels F, Guedes AC, Amaro HM, Kijjoa A, Vasconcelos V. Phycobiliproteins from cyanobacteria: Chemistry and biotechnological applications. Biotechnol Adv [Internet]. 2019 Feb 21 [cited 2019 Mar 11]; Available from: https://www.sciencedirect.com/science/article/pii/S0734975019300291
Ritala A, Häkkinen ST, Toivari M, Wiebe MG. Single cell protein-state-of-the-art, industrial landscape and patents 2001-2016. Frontiers in Microbiology. 2017.
Becker EW. Micro-algae as a source of protein. Biotechnology Advances. 2007.
Gao K, Ma Z. Photosynthesis and growth of Arthrospira (Spirulina) platensis (Cyanophyta) in response to solar UV radiation, with special reference to its minor variant. Environ Exp Bot. 2008;
Juneja A, Ceballos RM, Murthy GS. Effects of environmental factors and nutrient availability on the biochemical composition of algae for biofuels production: A review. Energies. 2013.
Panahi Y, Khosroshahi AY, Sahebkar A, Heidari HR. Impact of cultivation condition and media content on Chlorella vulgaris composition. Advanced Pharmaceutical Bulletin. 2019.
Ismaiel MMS, El-Ayouty YM, Piercey-Normore M. Role of pH on antioxidants production by Spirulina (Arthrospira) platensis. Brazilian J Microbiol. 2016;
Fields MW, Hise A, Lohman EJ, Bell T, Gardner RD, Corredor L, et al. Sources and resources: Importance of nutrients, resource allocation, and ecology in microalgal cultivation for lipid accumulation. Applied Microbiology and Biotechnology. 2014.
Fu L, Li Q, Yan G, Zhou D, Crittenden JC. Hormesis effects of phosphorus on the viability of Chlorella regularis cells under nitrogen limitation. Biotechnol Biofuels. 2019;
Shen XF, Liu JJ, Chu FF, Lam PKS, Zeng RJ. Enhancement of FAME productivity of Scenedesmus obliquus by combining nitrogen deficiency with sufficient phosphorus supply in heterotrophic cultivation. Appl Energy. 2015;
Tao R, Bair R, Lakaniemi AM, van Hullebusch ED, Rintala JA. Use of factorial experimental design to study the effects of iron and sulfur on growth of Scenedesmus acuminatus with different nitrogen sources. J Appl Phycol. 2019;
Yeesang C, Cheirsilp B. Effect of nitrogen, salt, and iron content in the growth medium and light intensity on lipid production by microalgae isolated from freshwater sources in Thailand. Bioresour Technol. 2011;
Ward VCA, Rehmann L. Fast media optimization for mixotrophic cultivation of Chlorella vulgaris. Sci Rep. 2019;
- Abstract Viewed: 639 times
- PDF Downloaded: 702 times
