Improvement of Astaxanthin Production in Coelastrum sp. by Optimization Using Taguchi Method
Applied Food Biotechnology,
Vol. 7 No. 4 (2020),
18 August 2020
,
Page 205-214
https://doi.org/10.22037/afb.v7i4.29697
Abstract
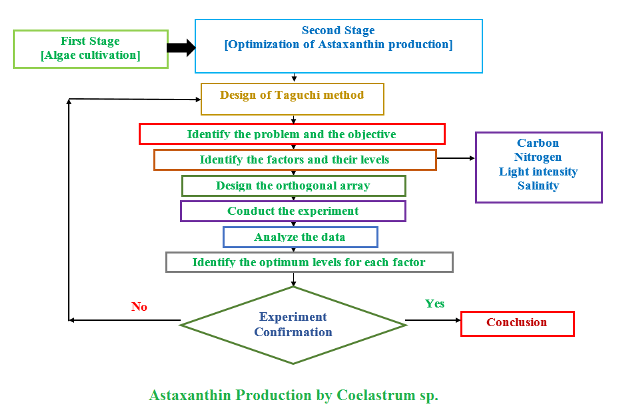
Background and Objective: Astaxanthin is a keto-carotenoid pigment known as one of the most valuable compounds with great potentials in the market. It has widely been used in nutraceutical, pharmaceutical, cosmetics and food industries due to its strong antioxidant activity. Green microalgae seem as promising natural sources in production of astaxanthin. The aim of this study was to optimize astaxanthin production in Coelastrum sp. to overcome low productivity of microalgae.
Materials and Methods: This study was carried out using experimentally statistical technique and Taguchi method to find optimum conditions for maximizing production of astaxanthin in green microalgae, Coelastrum sp. Effects of nutritional (carbon and nitrogen) and environmental (light and salinity) factors on biomass and astaxanthin production were investigated. Experiments were carried out for light intensity (250-550 µmol photons m-2 s-1), salinity using sodium chloride (1.0-3.0 g l-1), carbon source using sodium acetate
(0.5-2.0 g l-1) and nitrogen source using sodium nitrate (0.1-0.3 g l-1).
Results and Conclusion: Results showed that optimum conditions of astaxanthin production in Coelastrum sp. included 250 µmol photons m-2 s-1 of light intensity, 3 g l-1 salinity, 0.5 g l-1 carbon and 0.1 g l-1 nitrogen with a maximum yield of astaxanthin (14.44 mg l-1), which was 2-fold higher than that before optimization. This optimization resulted in high quantities of astaxanthin production using optimization of conditions that affected production yields of astaxanthin from Coelastrum sp.
- Astaxanthin
- Coelastrum sp.
- optimization
- production
- Taguchi method
How to Cite
References
Zhang W, Wang J, Liu, T. Attached cultivation of Haematococcus pluvialis for astaxanthin production. Bioresour Technol. 2014; 158: 329–335.
https://doi.org/10.1016/j.biortech.2014.02.044
Dufossé L, Galaupa P, Yaronb A, Arad SM, Blanc P, Chidambara MKN, Ravishankard GA. Microorganisms and microalgae as sources of pigments for food use: a scientific oddity or an industrial reality? Trends Food Sci. Technol. 2005; 16: 389– 406.
https://doi.org/10.1016/j.tifs.2005.02.006
Koller M, Muhr A, Braunegg G. Microalgae as versatile cellular factories for valued products. Algal Research. 2014; 6: 52-63.
https://doi.org/10.1016/j.algal.2014.09.002
Capelli B, Bagchi D, Cysewski G. Synthetic astaxanthin is significantly inferior to algal-based astaxanthin as an antioxidant and may not be suitable as a human nutritional supplement. Nutra Foods. 2013; 12: 145-152.
https://doi.org/10.1007/s13749-013-0051-5
Orosa M, Franqueira D, Cid A, Abalde J. Carotenoid accumulation in Haematococcus pluvialis in mixotrophic growth. Biotechnol Lett. 2001; 23: 373–378.
https://doi.org/10.1023/A:1005624005229
Liu J, Sun Z, Gerken H, Liu Z, Jiang Y, Chen F. Chlorella zofingiensis as an alternative microalgal Producer of astaxanthin: biology and industrial potential. Mar Drugs. 2014; 12: 3487-3515.
https://doi.org/10.3390/md12063487
Liu Z, Liu C, Hou Y, Chen S, Xiao D, Zhang J, Chen F. Isolation and characterization of a marine microalgae for biofuel production with astaxanthin as a co-product. Energies. 2013; 6(6): 2759-2772.
https://doi.org/10.3390/en6062759
Boussiba S. Carotenogensis in the green alga Haematococcus pluvialis: cellular physiology and stress response. Physiol. Plant. 2000; 108(2): 111-117.
https://doi.org/10.1034/j.1399-3054.2000.108002111.x
Fábregas J, Otero A, Maseda A, Domínguez A. Two-stage cultures for the production of astaxanthin from Haematococcus pluvialis. J. Biotechnol. 2001; 89(1): 65-71.
https://doi.org/10.1016/S0168-1656(01)00289-9
Beg QK, Sahai V, Gupta R. Statistical media optimization and alkaline protease production from Bacillus mojaensis in a bioreactor. Process Biochem. 2003; 39(2): 203–209.
https://doi.org/10.1016/S0032-9592(03)00064-5
Taguchi G. Introduction to Quality Engineering: Designing Quality into Products and Processes. Asian Productivity Organization, Tokyo. 1986.
Dasu VV, Panda T, Chidambaram M. Determination of significant parameters for improved griseofulvin production in a batch bioreactor by Taguchi’s method. Process Biochem. 2003; 38: 877–880.
https://doi.org/10.1016/S0032-9592(02)00068-7
Mohan NS, Ramachandra A, Kulkarni SM. Influence of process parameters on cutting force and torque during drilling of glass–fiber polyester reinforced composites. Composite Structures. 2005; 71(3-4): 407–413.
https://doi.org/10.1016/j.compstruct.2005.09.039
Hsia SY, Yang SK. Enhancing algal growth by stimulation with LED lighting and ultrasound. Journal of Nanomaterials. 2015; vol. 2015, Article ID 53352, 11 pages.
https://doi.org/10.1155/2015/531352
Guo X, Li X, Xiao D. Optimization of culture conditions for production of astaxanthin by Phaffia rhodozyma. 4th International Conference on Bioinformatics and Biomedical Engineering. 2010; 1-4.
DOI: 10.1109/ICBBE.2010.5516101
Chen Y, Mu C, Xavier I, Britton C. Signal-to-noise analysis for detection sensitivity of small absorbing heterogeneity in turbid media with single source and dual-interfering-source. Optic Express (OSA). 2001; 9(4): 212-224.
https://doi.org/10.1364/OE.9.000212
Boussiba S, Vonshak A. Astaxanthin accumulation in the green alga Haematococcus pluvialis. Plant Cell Physiol. 1991; 32(7): 1077–1082.
https://doi.org/10.1093/oxfordjournals.pcp.a078171
Brinda BR, Sarada R, Sandesh KB, Ravishankar GA. Accumulation of astaxanthin in flagellated cells of Haematococcus pluvialis-cultural and regulatory aspects. Curr. Sci. 2004; 87: 1290-1295.
Sarada R, Vidhyavathi R, Usha D, Ravishankar GA. An efficient method for extraction of astaxanthin from green algae Haematococcus pluvialis. J. Agric. Food Chem. 2006; 54(20): 7585-7588.
https://doi.org/10.1021/jf060737t
Escamilla EM, Dendooven L, Magana IP, Parra R, De la Torre M. Optimization of gibberellic acid production by immobilized Gibberella fujikuroi mycelium in fluidized bioreactors. J Biotech. 2000; 76(2-3): 147–155.
https://doi.org/10.1016/S0168-1656(99)00182-0
Roy RK. Design of experiments using the Taguchi approach: 16 Steps to Product and process improvement. John Wiley & Sons, Inc., United States. 2001.
Athreya S, Venkatesh D. Application of Taguchi method for optimization of process parameters in improving the surface roughness of lathe facing operation. International Refereed Journal of Engineering and Science (IRJES). 2012; 1(3): 13-19.
Sorana DB, Lorentz J. Design of Experiments: Useful Orthogonal Arrays for Number of Experiments from 4 to 16. Entropy. 2007; 9(4): 198-232.
https://doi.org/10.3390/e9040198
Tripathi U, Sarada R, Ravishankar G. Effect of culture conditions on growth of green alga-Haematococcus pluvialis and astaxanthin production. Acta Physiol. Plant. 2002; 24(3): 323-329.
https://doi.org/10.1007/s11738-002-0058-9
Saha SK, McHugh E, Hayes J, Moane S, Walsh D, Murray P. Effect of various stress-regulatory factors on biomass and lipid production in microalga Haematococcus pluvialis. Bioresour. Technol. 2013; 128: 118-124.
https://doi.org/10.1016/j.biortech.2012.10.049
Deas M, Orlob GK. River Modeling Project. 1999.
- Abstract Viewed: 838 times
- PDF Downloaded: 651 times
