Modified Explant Culture is the Effective Technique for Isolating Mesenchymal Stem Cells from Glioblastoma
School of Medicine Students' Journal,
Vol. 4 (2022),
28 November 2022
https://doi.org/10.22037/smsj.v4i1.38856
Abstract
Background and Aim: Until now, mesenchymal stem cells (MSCs) have been obtained from a variety of sources, including several tumor types. In order to acquire cells with the best quality and function while also taking economic factors into account, selecting an appropriate isolation procedure is a crucial step. The goal of this study was to use a modified explant culture approach to isolation of pure mesenchymal stem cells from human glioblastoma.
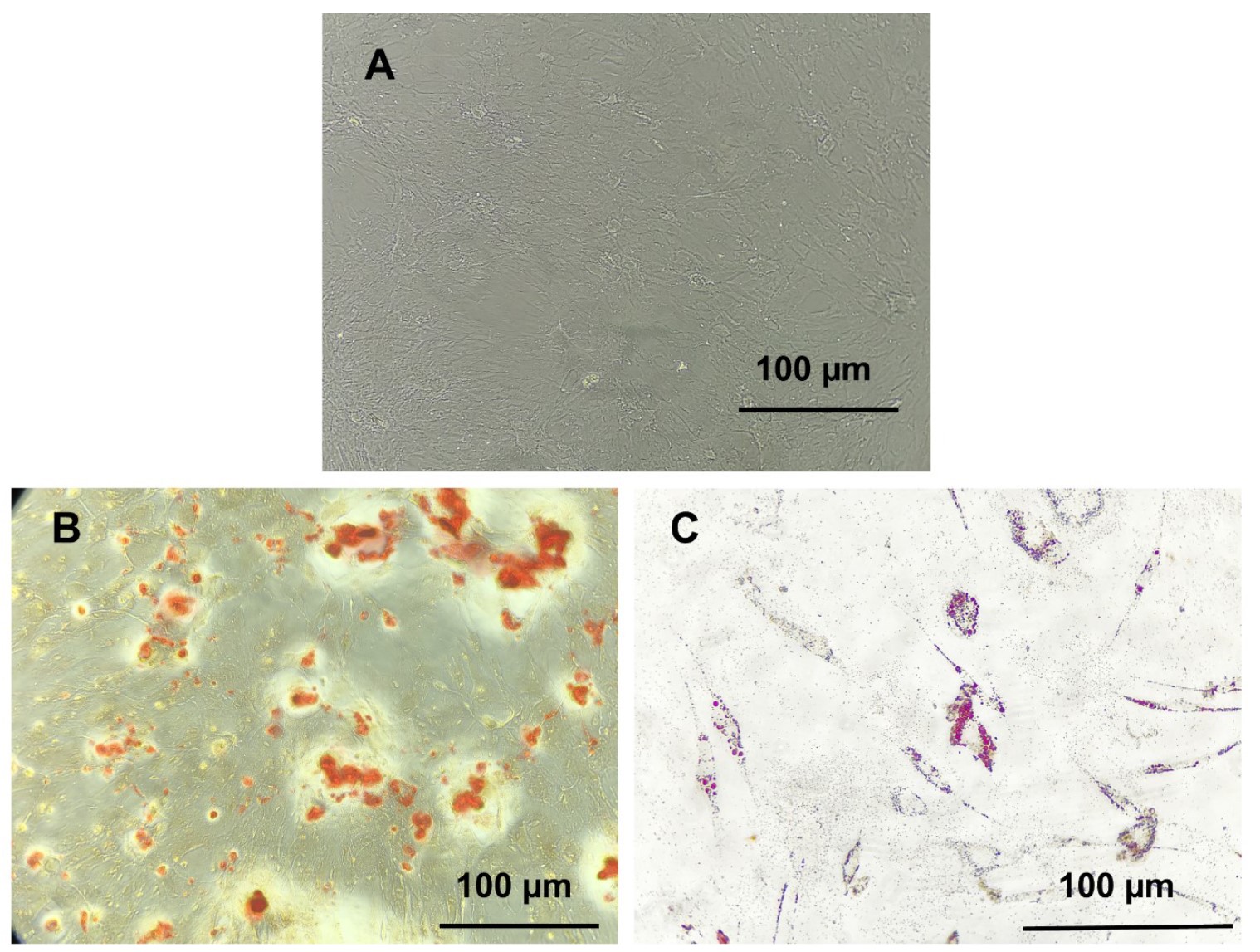
Methods: Six samples of glioblastoma tumors were cut into cube - like fragments of 1-3 mm in diameter (explants). The explants were put into the wells of a 24-well plate, cultured in Dulbecco's Modified Eagle's medium (DMEM), and kept at 37°C with 5% CO2 in an incubator. Inverted microscopy was used to analyze the morphological phenotypes of the cells. Wells with uniform fibroblast-like cell morphology were considered positive and chosen for further expansion and identification.
Results: Totally, 38.46% of the wells were chosen for development after testing positive for all grown explants. According to flow cytometry analyses, the isolated cells were positive for CD73, CD105, and CD90 but negative for CD45 and CD34. The cells also successfully underwent multipotential differentiation into adipocytes and osteoblasts.
Conclusion: Overall, our research demonstrated that GA - MSC could successfully be isolated from human Glioblastoma tumor tissues utilizing a modified explant culture method. With this approach, we were able to rapidly and efficiently develop a more pure MSC population by choosing tumor pieces with the potential to produce a population of homogenous cells with fibroblast - like morphology and removing pieces whose heterogeneous cells were moved. The approach is also workable and affordable. The results might serve as a framework for more investigation into GA - MSCs, glioblastoma TME, and their mechanism of development.
- Mesenchymal stem cells
- Explant culture
- Glioblastoma
- Tissue Culture Technique
- glioma-associated mesenchymal stem cells
How to Cite
References
Tan AC, Ashley DM, López GY, Malinzak M, Friedman HS, Khasraw M. Management of glioblastoma: State of the art and future directions. CA Cancer J Clin. 2020;70(4):299–312.
Yao M, Li S, Wu X, Diao S, Zhang G, He H, et al. Cellular origin of glioblastoma and its implication in precision therapy. Cell Mol Immunol. 2018;15(8):737–9.
Chinopoulos C, Seyfried TN. Mitochondrial Substrate-Level Phosphorylation as Energy Source for Glioblastoma: Review and Hypothesis. ASN Neuro [Internet]. 2018 Dec 1 [cited 2022 Jul 7];10. Available from: https://journals.sagepub.com/doi/full/10.1177/1759091418818261
Wu W, Klockow JL, Zhang M, Lafortune F, Chang E, Jin L, et al. Glioblastoma multiforme (GBM): An overview of current therapies and mechanisms of resistance. Pharmacol Res. 2021 Sep 1;171:105780.
Garnier D, Ratcliffe E, Briand J, Cartron PF, Oliver L, Vallette FM. The Activation of Mesenchymal Stem Cells by Glioblastoma Microvesicles Alters Their Exosomal Secretion of miR-100-5p, miR-9-5p and let-7d-5p. Biomed 2022, Vol 10, Page 112 [Internet]. 2022 Jan 6 [cited 2022 Jul 6];10(1):112. Available from: https://www.mdpi.com/2227-9059/10/1/112/htm
Dvorak HF. Tumors: Wounds That Do Not Heal—Redux. Cancer Immunol Res [Internet]. 2015 Jan 1 [cited 2022 Jul 6];3(1):1–11. Available from: https://aacrjournals.org/cancerimmunolres/article/3/1/1/467415/Tumors-Wounds-That-Do-Not-Heal-ReduxTumors-as
Pillat MM, Oliveira-Giacomelli Á, das Neves Oliveira M, Andrejew R, Turrini N, Baranova J, et al. Mesenchymal stem cell-glioblastoma interactions mediated via kinin receptors unveiled by cytometry. Cytom Part A [Internet]. 2021 Feb 1 [cited 2022 Jul 6];99(2):152–63. Available from: https://onlinelibrary.wiley.com/doi/full/10.1002/cyto.a.24299
Lou Q, Zhao M, Xu Q, Xie S, Liang Y, Chen J, et al. Retinoic Acid Inhibits Tumor-Associated Mesenchymal Stromal Cell Transformation in Melanoma. Front Cell Dev Biol. 2021;9(April):1–12.
Zippel N, Schulze M, on ET-R patents, 2010 undefined. Biomaterials and mesenchymal stem cells for regenerative medicine. ingentaconnect.com [Internet]. [cited 2022 Jul 7]; Available from: https://www.ingentaconnect.com/content/ben/biot/2010/00000004/00000001/art00001
Stefańska K, Mehr K, Wieczorkiewicz M, Kulus M, Angelova Volponi A, Shibli JA, et al. Stemness Potency of Human Gingival Cells—Application in Anticancer Therapies and Clinical Trials. Cells 2020, Vol 9, Page 1916 [Internet]. 2020 Aug 18 [cited 2022 Jul 7];9(8):1916. Available from: https://www.mdpi.com/2073-4409/9/8/1916/htm
Lin W, Huang L, Li Y, Fang B, Li G, … LC-BR, et al. Mesenchymal stem cells and cancer: clinical challenges and opportunities. hindawi.com [Internet]. [cited 2022 Jul 7]; Available from: https://www.hindawi.com/journals/bmri/2019/2820853/
Sineh Sepehr K, Razavi A, Saeidi M, Mossahebi-Mohammadi M, Abdollahpour-Alitappeh M, Hashemi SM. Development of a novel explant culture method for the isolation of mesenchymal stem cells from human breast tumor. J Immunoass Immunochem [Internet]. 2018;39(2):207–17. Available from: https://doi.org/10.1080/15321819.2018.1447487
Papait A, Stefani FR, Cargnoni A, Magatti M, Parolini O, Silini AR. The Multifaceted Roles of MSCs in the Tumor Microenvironment: Interactions With Immune Cells and Exploitation for Therapy. Front Cell Dev Biol. 2020;8(June):1–13.
Kucerova L, Matuskova M, Hlubinova K, Altanerova V, Altaner C. Tumor cell behaviour modulation by mesenchymal stromal cells. Mol Cancer. 2010;9:1–15.
Kim SH, Choe C, Shin YS, Jeon MJ, Choi SJ, Lee J, et al. Human lung cancer-associated fibroblasts enhance motility of non-small cell lung cancer cells in co-culture. Anticancer Res. 2013;33(5):2001–9.
Valente MJ, Henrique R, Costa VL, Jerónimo C, Carvalho F, Bastos ML, et al. A rapid and simple procedure for the establishment of human normal and cancer renal primary cell cultures from surgical specimens. PLoS One. 2011;6(5):21–3.
Hendijani F. Explant culture: An advantageous method for isolation of mesenchymal stem cells from human tissues. Cell Prolif. 2017;50(2):1–14.
Nicodemou A, Danisovic L. Mesenchymal stromal/stem cell separation methods: concise review. Cell Tissue Bank. 2017;18(4):443–60.
Priya N, Sarcar S, Majumdar A Sen, Sundarraj S. Explant culture: a simple, reproducible, efficient and economic technique for isolation of mesenchymal stromal cells from human adipose tissue and lipoaspirate. J Tissue Eng Regen Med [Internet]. 2014 [cited 2022 Jul 10];8(9):706–16. Available from: https://pubmed.ncbi.nlm.nih.gov/22837175/
D’Souza N, Burns JS, Grisendi G, Candini O, Veronesi E, Piccinno S, et al. MSC and Tumors: Homing, Differentiation, and Secretion Influence Therapeutic Potential. Adv Biochem Eng Biotechnol [Internet]. 2013 [cited 2022 Jul 10];130:209–66. Available from: https://pubmed.ncbi.nlm.nih.gov/22990585/
Brune JC, Tormin A, Maria CJ, Rissler P, Brosjö O, Löfvenberg R, et al. Mesenchymal stromal cells from primary osteosarcoma are non-malignant and strikingly similar to their bone marrow counterparts. Int J cancer [Internet]. 2011 Jul 15 [cited 2022 Nov 3];129(2):319–30. Available from: https://pubmed.ncbi.nlm.nih.gov/20878957/
Lee DH, Joo SD, Han SB, Im J, Lee SH, Sonn CH, et al. Isolation and expansion of synovial CD34(-)CD44(+)CD90(+) mesenchymal stem cells: comparison of an enzymatic method and a direct explant technique. Connect Tissue Res [Internet]. 2011 Jun [cited 2022 Jul 10];52(3):226–34. Available from: https://pubmed.ncbi.nlm.nih.gov/21117906/
Spath L, Rotilio V, Alessandrini M, Gambara G, De Angelis L, Mancini M, et al. Explant-derived human dental pulp stem cells enhance differentiation and proliferation potentials. J Cell Mol Med [Internet]. 2010 Jun [cited 2022 Jul 10];14(6B):1635–44. Available from: https://pubmed.ncbi.nlm.nih.gov/19602052/
Ishige I, Nagamura-Inoue T, Honda MJ, Harnprasopwat R, Kido M, Sugimoto M, et al. Comparison of mesenchymal stem cells derived from arterial, venous, and Wharton’s jelly explants of human umbilical cord. Int J Hematol [Internet]. 2009 Sep [cited 2022 Jul 10];90(2):261–9. Available from: https://pubmed.ncbi.nlm.nih.gov/19657615/
Otte A, Bucan V, Reimers K, Hass R. Mesenchymal stem cells maintain long-term in vitro stemness during explant culture. Tissue Eng Part C Methods [Internet]. 2013 Dec 1 [cited 2022 Jul 10];19(12):937–48. Available from: https://pubmed.ncbi.nlm.nih.gov/23560527/
- Abstract Viewed: 93 times
- PDF Downloaded: 13 times




