Production, Recovery and Characterization of an Enterocin with Anti-Listerial Activity Produced by Enterococcus hirae OS1
Applied Food Biotechnology,
Vol. 7 No. 2 (2020),
10 March 2020
,
Page 103-114
https://doi.org/10.22037/afb.v7i2.27582
Abstract
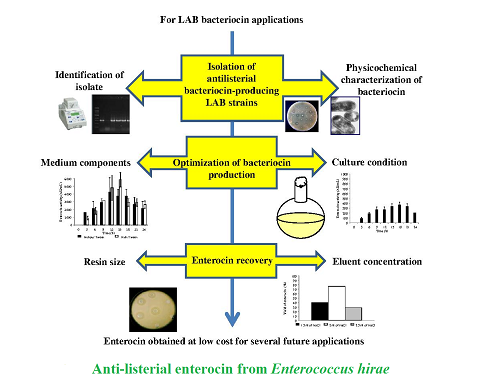
Background and objective: Lactic acid bacteria, used in food processing for a long time, can produce various metabolites during their growth, including bacteriocins. These antimicrobials, used as natural bio-preservatives, enhance the food safety. The objective of this study was to assess lactic acid bacteria bacteriocins with anti-listerial activity and optimize their production and recovery process.
Material and methods: Isolate was identified using conventional assays (morphological and biochemical characteristics) and 16S rRNA gene sequencing. Lactic acid bacteria bacteriocins were characterized based on their physicochemical properties (nature, pH stability and thermo-resistance). The production process was based on optimization of media components (growth media and addition of glucose, nitrogen source and tween 20) and culture conditions (temperature, pH, agitation and inoculum size). Furthermore, optimization of the recovery process was studied using ion exchange chromatography on amberlite IRC-50 (effects of resin size and NaCl eluent concentration).
Results and conclusion: Enterococcus hirae OS1 was isolated from Moroccan raw cow milk as bacteriocinogenic strain. After optimization of the bacteriocin production process, results showed that the key parameters for increasing of production included temperature of 30°C, pH of 6.5 and inoculum size of 5%. Production with whey-based and economic food-grade substrate allowed high production of enterocin OS1 (1,600 AU ml-1) (P<0.001). Simultaneous addition of glucose (1%) and Tween 20 (1%) increased enterocin titer significantly (5,866 AU ml-1) (P<0.01). Recovery efficiency increased with use of 70% amberlite IRC-50 resin and elution with 2M NaCl. Indeed, recovery of 75.4% of bacteriocin was achieved in comparison to 15.7% of bacteriocin without optimization. This promises achieving high quantities of enterocin at low costs.
Conflict of interest: The authors declare no conflict of interest.
- ▪ Enterocin ▪ Enterococcus hirae ▪ Lactic acid bacteria ▪ Listeria monocytogenes ▪ Optimization ▪ Whey
How to Cite
References
EFSA (European Food Safety Authority) and ECDC (European Centre for Disease Prevention and Control), 2016. The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2015. EFSA Journal, 14(12): 4634, 231 pp. DOI:10.2903/j.efsa.2016.4634.
CAPM, Centre Anti Poison et de Pharmacovigilance du Maroc. 2015. Maladies d’origine alimentaire. Toxicologie Maroc 27 - 3ème trimestre.
Stiles, M.E. 1996. Biopreservation by lactic acid bacteria. Antonie Van Leeuwenhoek, 70: 331-345. DOI: 10.1007/BF00395940
Ananou, S., Maqueda, M., Martínez-Bueno, M. and Valdivia, E. 2007. Biopreservation, an ecological approach to improve the safety and shelf-life of foods. In: Communicating Current Research and Educational Topics and Trends in Applied Microbiology. pp. 475-486. Vol. 1. Editorial Formatex, Badajoz. ISBN 978-84-611-9423-0.
Mayr-Harting, A., Hedges, A.J. and Berkeley, R.C.W. 1972. Methods for studying bacteriocins. In: Methods in Microbiology. Norris, J.R. and Ribbons, D.W. (ed.). pp. 315-342. Academic Press. New York.
Gálvez, A., Maqueda, M., Valdivia, E., Quesada, A., and Montoya, E. 1986. Characterization and partial purification of a broad spectrum antibiotic AS-48 produced by Streptococcus faecalis. Canadian Journal of Microbiology, 32: 765-771. DOI: 10.1139/m86-141
Gomori G, 1955. Preparation of Buffers for Use in Enzyme Active Studies. In: Methods in Enzymology, Colwick, S.P. and N.O. Kaplan (Eds.). Vol. I, Academic Press, New York.
Stiles, M.E., and Holzapfel, W.H. 1997. Lactic acid bacteria of foods and their current taxonomy. International Journal of Food Microbiology, 36: 1-29. DOI: 10.1016/s0168-1605(96)01233-0
Carr, F.J., Chill, D. and Maida, N. 2002. The lactic acid bacteria: A literature survey. Critical Reviews in Microbiology, 28: 281-370. DOI: 10.1080/1040-840291046759
Martín-Platero, A. M., Valdivia, E., Maqueda, M., Martínez-Bueno, M. 2007. Fast, convenient, and economical method for isolating genomic DNA from lactic acid bacteria using a modification of the protein "salting-out" procedure. Analytical Biochemistry, 366: 102-104. DOI: 10.1016/j.ab.2007.03.010
Weisberg, W.G., Barns, S.M., Pelletier, D.A. and Lane, D.J. 1991. 16S ribosomal DNA amplification for phylogenetic study. Journal of Bacteriology, 173: 697-703. DOI: 10.1128/jb.173.2.697-703.1991
Ananou, S., Muñoz, A., Gálvez, A., Martínez-Bueno, M., Maqueda, M. and Valdivia, E. 2008. Optimization of enterocin AS-48 production on a whey-based substrate. International Dairy Journal, 18: 923-927. DOI: 10.1016/j.idairyj.2008.02.001
Parente, E. and Ricciardi, A. 1994. Influence of pH on the production of enterocin 1146 during batch fermentation. Letters in Applied Microbiology, 19: 12-15. DOI: 10.1111/j.1472-765X.1994.tb00891.x
Cleveland, J., Montville, T.J., Nes, I.F. and Chikindas, M.L. 2001. Bacteriocins: safe, natural antimicrobials for food preservation. International Journal of Food Microbiology, 7: 1-20. DOI: 10.1016/S0168-1605(01)00560-8
Luana, R.F., Bruna, R.B.P., Eliezer, M.P.E. and Janaína, D.S.N. 2011. Influence of growth conditions on production of Klebicin K and Raoultellin L, two antimicrobial substances against gram-negative pathogens. International Journal of Pharmaceutical and Biological Archives, 2: 1095-1099.
Khedid, K., Faid, M., Mokhtari, A., Soulaymani, A., and Zinedine, A. 2009. Characterization of lactic acid bacteria isolated from the one humped camel milk produced in Morocco. Microbiological Research, 164: 81-91. DOI: 10.1016/j.micres.2006.10.008
Achemchem, F., Cebrián, R., Abrini, J., Martínez-bueno, M., Valdivia, E. and Maqueda, M. 2012. Antimicrobial characterization and safety aspects of the bacteriocinogenic Enterococcus hirae F420 isolated from Moroccan raw goat milk. Canadian Journal of Microbiology, 58: 596-604. DOI: 10.1139/w2012-027
Giraffa, G. 2014. Enterococcus. In: Encyclopedia of Food Microbiology (Second Edition). Batt, C.A. and Tortorello, M.L. (Eds). Pp. 674-679. Academic Press. ISBN: 9780123847331.
Cavicchioli, V.Q., Camargo, A.C., Todorov, S.D. and Nero L.A. 2017. Novel bacteriocinogenic Enterococcus hirae and Pediococcus pentosaceus strains with antilisterial activity isolated from Brazilian artisanal cheese. Journal of Dairy Science, 100: 2526-2535. DOI: 10.3168/jds.2016-12049
Abriouel, H., Valdivia, E., Martínez-Bueno, M., Maqueda M., and Gálvez, A. 2003. A simple method for semi-preparative-scale production and recovery of enterocin AS-48 derived from Enterococcus faecalis subsp. liquefaciens A-48-32. Journal of Microbiological Methods, 55: 599-605. DOI: 10.1016/S0167-7012(03)00202-1
Morgan, S.M., Galvin, M., Kelly, J., Ross, R.P. and Hill, C. 1999. Development of a lacticin 3147-enriched whey powder with inhibitory activity against foodborne pathogens. Journal of Food Protection, 62, 1011-1016. DOI: 10.4315/0362-028X-62.9.1011
Huggenholtz, J., and De Veer, G.J.C.M. 1991. Application of nisin A and nisin Z in dairy technology. In: Nisin and novel lantibiotics. G. Jung, G. and H-G. Sahl, H-G. (Eds.), pp. 440-447. Leiden, The Netherlands, Leiden: Escom Sciences Publishers B.V.
Amiali, M.N., Lacroix, C., and Simard, R.E. 1998. High nisin Z production by Lactococcus lactis subsp. lactis biovar. diacetylactis UL719 in whey permeate with aeration. World Journal of Microbiology and Biotechnology, 14: 887-894. DOI: 10.1023/A:1008863111274
Dicks, L.M.T., Dreyer, L., Smith, C. and van Staden, A.D. 2018. A review: The fate of bacteriocins in the human gastro-intestinal tract: do they cross the gut-blood barrier?. Front Microbiology, 9: 2297. DOI: 10.3389/fmicb.2018.02297
De Vuyst, L., Callewaert, R., and Crabbé, K. 1996. Primary metabolite kinetics of bacteriocin biosynthesis by Lactobacillus amylovorus and evidence for stimulation of bacteriocin production under unfavourable growth conditions. Microbiology, 142, 817-827. DOI: 10.1099/00221287-142-4-817
Kim, M-H., Kong, Y-J., Baek, H., and Hyun, H-H. (2006). Optimization of culture conditions and medium composition for the production of micrococcin GO5 by Micrococcus sp. GO5. Journal of Biotechnology, 121: 54–61. DOI: 10.1016/j.jbiotec.2005.06.022
Sahar, A., Ramakrishnan, N.R., Tengku, A.T.I, Shuhaimi, M., Rosfarizan, M., Hassan, M.D. and Arbakariya, B.A. 2011. Effect of medium composition and culture condition on the production of bacteriocin-like inhibitory substances (blis) by Lactobacillus paracasei la07, a strain isolated from budu. Biotechnology and Biotechnological Equipment, 4 : 2652-2657. DOI: 10.5504/BBEQ.2011.0101
Baldasso, C., Barros, T.C. and Tessaro, I.C. 2011. Concentration and purification of whey proteins by ultrafiltration. Desalination, 278: 381–386. DOI: 10.1016/j.desal.2011.05.055
Von Mollendorff, J.W., Todorov, S.D. and Dicks, L.M.T. 2009. Optimization of growth medium for production of bacteriocins produced by Lactobacillus plantarum JW3BZ and JW6BZ, and Lactobacillus fermentum JW11BZ and JW15BZ isolated from Boza. Trakia Journal of Sciences, 7: 22-33.
Castro, M.P., Palavecino, N.Z., Herman C., Garro, O.A. and Campos, C.A. 2011. Lactic acid bacteria isolated from artisanal dry sausages: Characterization of antibacterial compounds and study of the factors affecting bacteriocin production. Meat Science, 87: 321-329. DOI: 10.1016/j.meatsci.2010.11.006
Chan-Ick, C., Moo-Chang, K., Seong-Bo, K., Young-Ho, H. and Yu-Ryang, P. 2004. Simple one-step purification of nisin Z from unclarified culture broth of Lactococcus lactis subsp. Lactis A164 using expanded bed ion exchange chromatography. Biotechnology Landters, 26: 1341–1345. DOI: 10.1023/B:BILE.0000045630.29494.45
Achemchem F., Martinez-Bueno M., Abrini J., Valdivia E. and Maqueda M., 2005. Enterococcus faecium F58, a bacteriocinogenic strain naturally occurring in Jben, a soft, farmhouse goat's cheese made in Morocco. Journal of Applied Microbiology, 99: 141-150. DOI: 10.1111/j.1365-2672.2005.02586.x
Ennahar, S., Asou, Y., Zendo, T., Sonomoto, K. and Ishizaki, A., 2001. Biochemical and genetic evidence for production of enterocins A and B by Enterococcus faecium WHE 81. International Journal of Food Microbiology, 70 : 291–301. DOI: 10.1016/s0168-1605(01)00565-7
Gutierrez J., Criado R., Martin M., Herranz C., Cintas L.M. and Hernandez P.E. 2005. Production of enterocin P, an antilisterial pediocin-like bacteriocin from Enterococcus faecium P13, in Pichia pastoris. Antimicrobial Agents and Chemotherapy, 49: 3004-3008. DOI: 10.1128/AAC.49.7.3004-3008.2005
Li, Y., Xiang, H., Liu, J., Zhou, M. and Tan, H. 2003. Purification and biological characterization of halocin C8, a novel peptide antibiotic from Halobacterium strain AS7092. Extremophiles, 7: 401-407. DOI: 10.1007/s00792-003-0335-6
- Abstract Viewed: 561 times
- PDF Downloaded: 706 times