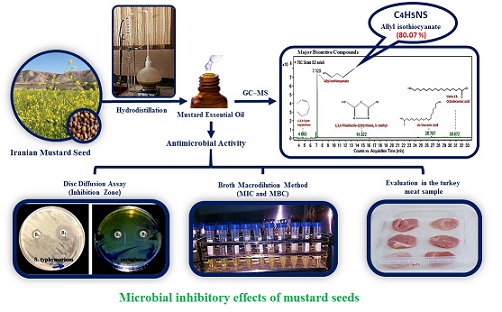
Comparison of the Chemical Compositions and Antibacterial Activities of Two Iranian Mustard Essential Oils and Use of these Oils in Turkey Meats as Preservatives
Applied Food Biotechnology,
Vol. 6 No. 4 (2019),
24 September 2019
,
Page 225-236
https://doi.org/10.22037/afb.v6i4.25687
Abstract
Background and objective: Iranian mustard is cultivated in southern areas of Iran and used traditionally as natural preservative. Aims of the current study were identification and comparison of the chemical compositions and antibacterial activities of two Iranian mustard essential oils and assessment of these oils use for increasing the shelf life of turkey meats.
Material and methods: Chemical compositions of two Iranian mustard essential oils were identified using gas chromatography-mass spectrometry and antibacterial activities of these oils were assessed against Salmonella typhimurium, Escherichia coli, Citrobacter freundii, Pseudomonas aeruginosa, Staphylococcus aureus, Listeria monocytogenes, Bacillus cereus and Enterococcus faecalis using disc diffusion and broth macrodilution assays. Inhibitory effects of the essential oils were assessed on growth of mesophilic psychrotrophic bacteria, yeasts and molds and sensory evaluation was carried out for the turkey meats.
Results and conclusion: Results of GC-MS showed presence of bioactive constituents, especially allyl isothiocyanate (75.87-80.07%). All the bacterial growth, especially for Escherichia coli, was inhibited with inhibition zones of greater than 20 mm and minimum inhibitory and bactericidal concentrations of 0.156 mg ml-1. Treatment of turkey meat samples with the mustard essential oils significantly decreased the count of mesophilic psychrotrophic bacteria, yeasts and molds during 20 days of storage at 4°C ±1, compared to controls (P≤0.05). Over the time, the sensory score of the treated samples increased, compared to controls. Based on these findings, the Iranian mustard essential oils can be used as natural preservatives in foods.
Conflict of interest: The authors declare no conflict of interest.- ▪ Allyl isothiocyanate ▪ Antimicrobial activity ▪ Foodborne bacteria ▪ Mustard essential oil ▪ Turkey meat
How to Cite
References
Reyes-Jurado F, Lo´pez-Malo A, Palou E. Antimicrobial Activity of Individual and Combined Essential Oils against Foodborne Pathogenic Bacteria. J Food Prot. 2016; 79: 309 –315.
Kumar D, Tanwar V K. Effects of incorporation of ground mustard on quality attributes of chicken nuggets. J Food Sci Techno. 2011; 48: 759–762.
Abul-Fadl MM, El-Badry N, and Ammar MS. Nutritional and Chemical Evaluation for Two Different Varieties of Mustard Seeds. J. World Appl. Sci. 2011; 15: 1225-1233.
Adeli Milani M, Mizani M, Ghavami M, Eshratabadi P. Comparative Analysis of Antimicrobial Properties of Mustard Paste and Powder in Mayonnaise. Eur. J. Biol. Res. 2014; 4: 412-418.
Zarei M, Najafzadeh H, Eskandari M H, Pashmforoush M, Enayati A, Gharibi D, Fazlara A. Chemical and microbial properties of mahyaveh, a traditional Iranian fish sauce. Food Control. 2012; 23: 511–514.
Yu J C, Jiang ZT, Li R, Chan S M. Chemical Composition of the Essential Oils of Brassica juncea (L.) Coss. Grown in Different Regions, Hebei, Shaanxi and Shandong, of China.J Food Drug Anal. 2003; 11:22-26.
Cui H, Baia M, Sunb Y, Abdel-Shafi Abdel-Samiea M, Lina L. Antibacterial activity and mechanism of Chuzhou chrysanthemum essential oil. J Funct Foods. 2018; 48:159–166.
NCCLS/CLSI - National Committee for Clinical Laboratory Standards. (2006). Performance standards for antimicrobial disk susceptibility tests. Approved standard, document M2-A9
Sarrazin S L F, Oliveira R B, Barata L E S, Mourão R H V. Chemical composition and antimicrobial activity of the essential oil of Lippia grandis Schauer (Verbenaceae) from the western Amazon. Food Chem. 2012; 134: 1474–1478.
Tendencia, E. A. (2004). Disk diffusion method. In Laboratory manual of standardized methods for antimicrobial sensitivity tests for bacteria isolated from aquatic animals and environment, 13-29.
Vetas D, Dimitropoulou E, Mitropoulou G, Kourkoutas Y, Giaouris E. Disinfection efficiencies of sage and spearmint essential oils against planktonic and biofilm Staphylococcus aureus cells in comparison with sodium hypochlorite. Int J Food Microbiol. 2017; 257:19-25.
Keivani Nahr F, Ghanbarzadeh B, Hamishehkar H, Samadi Kafil H. Food grade nanostructured lipid carrier for cardamom essential oil: Preparation, characterization and antimicrobial activity. J Funct Foods. 2018; 23: 412–422.
Fernández-Pan I, Carrión-Granda X, Maté, J I, Antimicrobial efficiency of edible coatings on the preservation of chicken breast fillets. Food Cont.2014; 36: 69-75.
Petrou S, Tsiraki M, Giatrakou M, Savvaidis I N. Chitosan dipping or oregano oil treatments, singly or combined on modified atmosphere packaged chicken breast meat. Int J Food Microbiol. 2012; 156: 264–271.
Yu D, Jiang Q, Xu Y, Xia W. The shelf life extension of refrigerated grass carp (Ctenopharyngodon idellus) fillets by chitosan coating combined with glycerol monolaurate.; Int. J. Biol. Macromol. 2017; 101:448–454.
Yun J, Fan X, Li X. Inactivation of Salmonella enterica serovar Typhimurium and quality maintenance of cherry tomatoes Treated with gaseous essential oils. J. Food Sci. 2013: 78: 458–464.
Romeo L, Iori R, Rollin P, Bramanti P, Mazzon E. Isothiocyanates: An Overview of Their Antimicrobial Activity against Human Infections. Molecules. 2018; 23: 624.
Bhattacharya ALY, Wade KL, Paonessa JD, Fahey JW, Zhang Y. Allyl isothiocyanate-rich mustard seed powder inhibits bladder cancer growth and muscle invasion. Carcinogenesis. 2012; 31: 2105–2110.
Turgis M, Han J, Caillet S, Lacroix, M. Antimicrobial activity of mustard essential oil against Escherichia coli O157:H7 and Salmonella Typhimorium. J Food cont. 2009; 20: 1073–1079.
Aparna V, Dileep KV, Mandal PK, Karthe P,Sadasivan C, Haridas M. Anti-inflammatory property of n-hexadecanoic acid: Structural evidence and kinetic assessment. CHEM BIOL DRUG DES. 2012; 80, 434–439.
Sharma A, Rai P K, Prasad S. GC–MS detection and determination of major volatile compounds in Brassica juncea L. leaves and seeds. Microchem J. 2018; 138: 488–493.
Zhou Z, Huang J, Hao H, Wei H, Zhou Y, Peng J. Applications of new functions for inducing host defense peptides and synergy sterilization of medium chain fatty acids in substituting in-feed antibiotics. J Funct Foods. 2019; 52: 348-359.
Mahadkar S, Valvi S, Jadhav V. Gas chromatography mass spectroscopic (GC-MS) analysis of some bioactive compounds from five medicinally relevant wild edible plants. J Food Prot. 2013; 63: 25–30.
Kabara J J, Swieczkowski D M, Conley A J, Truant J P. Fatty acids and derivatives as antimicrobial agents. Antimicrob Agents Chemother. 1972; 2: 23–28.
Galbraith H, MillerT B, Paton A M and Thompson J K. Antibacterial activity of long chain fatty acids and the reversal with calcium, magnesium, ergocalciferol and cholesterol. J Appl Bacteriol. 1971; 34:803–813.
Field C J, Blewett H H, Proctor S, Vine D. Human health benefits of vaccenic acid. Appl Physiol Nutr Metab. 2009; 34:979–991.
Mujeeb F, Bajpai P, Pathak N. Phytochemical Evaluation, Antimicrobial Activity, and Determination of Bioactive Components from Leaves of Aegle marmelos. BioMed Res Int, Article ID 497606. 2014; 11.
Tangjitjaroenkun J. Evaluation of antioxidant, antibacterial, and gas chromatography –mass spectrometry analysis of ethyl acetate extract of Streptomyces omiyansis SCH2. Asian J Pharm Clin Res. 2018; 11: 271-276.
Singh S, Das SS, Singh G, Perroti M, Schuff C, Catalán C A. Comparison of chemical composition, antioxidant and antimicrobial potentials of essential oils and oleoresins obtained from seeds of Brassica juncea and sinapis Alba. Moj Food Proc Technol. 2017; 4: 113‒120.
Bertoli A, Cirak C, Silva JAT. Hypericum species as sources of Valuable essential oils. Med Aromat Plant Sci Biotechnol. 2011; 5: 29-47.
Verkerk R, Schreiner M, Krumbein A, Ciska E, Holst B, Rowland I, De Schrijver R, Hansen M, Gerhauser C, Mithen R, Dekker M. Glucosinolates in Brassica vegetables: the influence of the food supply chain on intake, bioavailability and human health. Mol Nutr Food Res. 2009; 53: S219.
Akkoyun HT, Dostbil N, Kiran Ö. Antimicrobial Activity of Black Mustard Seed (Brassica nigra L.). Asian J. Chem. 2007; 19: 5533-5536.
Nazer A I, Kobilinsky A, Tholozana J L, Dubois-Brissonneta F. Combinations of food antimicrobials at low levels to inhibit the growth of Salmonella sv. Typhimurium: a synergistic effect. Food Microbiol. 2005; 22: 391-39.
Sharma A, Kumar A, Meena HS, Singh D. Chromatographic determination of phenolics in Brassica juncea L. Asian J Chem. 2017; 29: 296–300.
Luciano F B, Belland J, Holley R A. Microbial and chemical origins of the bactericidal activity of thermally treated yellow mustard powder toward Escherichia coli O157:H7 during dry sausage ripening. Int J Food Microbiol.2011; 145: 69–74.
Kim S A, Rhee M S. Highly en hanced bactericidal effects of medium chain fatty acids (caprylic, capric, and lauric acid) combined with edible plant essential oils (carvacrol, eugenol, beta-resorcylic acid, trans-cinnamaldehyde, thymol, and vanillin) against Escherichia coli O157:H7. Food Cont, 2016; 60: 447–454
Desbois A P, Smith V J. Antibacterial free fatty acids: Activities, mechanisms of action and biotechnological potential. Appl Microbiol Biotechnol. 2010; 85: 1629–1642.
. Adeli Milani M, Mizani M, Ghavami M, Eshratabadi P. (2013) The Physico-Chemical Influences of Yellow Mustard Paste - Comparison with the Powder in Mayonnaise. J Food Proc Technol.2014; 4: 210.
Shen Q, Wang M, Tian J, Hu, Ren S, Chen J, Ye X, Liu D. Effects of Chinese pickled and dried mustard on nutritional quality, sensory quality, and shelf life of steamed pork belly. Food Sci Nutr;2018;6(4):747-756.
- Abstract Viewed: 764 times
- PDF Downloaded: 777 times