Development of Synbiotic Milk Chocolate Enriched with Lactobacillus paracasei, D-tagatose and Galactooligosaccharide
Applied Food Biotechnology,
Vol. 5 No. 2 (2018),
23 March 2018
,
Page 59-68
https://doi.org/10.22037/afb.v5i2.19955
Abstract
Background and Objective: Prebiotics are food ingredients that induce the growth or activity of beneficial bacteria (Bifidobacteria and Lactobacilli). Galactooligosaccharide and tagatose are two main prebiotic compounds which are used in the food industry. Chocolate is widely consumed all over the world and could be used as an excellent vehicle for delivery of prebiotics. Furthermore, the incorporation of probiotics into chocolate, allows broadening the health claims of chocolate. The aim of the current study was to investigate the effect of tagatose and galactooligosaccharide on the physicochemical and sensory properties of milk chocolate and the survivability of Lactobacillus paracasei in the optimized formulation.
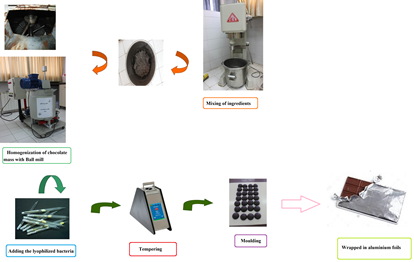
Material and Methods: Probiotic milk chocolate containing Lactobacillus paracasei were formulated by replacing a portion of the sucrose with the galactooligosaccharide powder and tagatose. For this purpose various concentrations of galactooligosaccharide and tagatose (2.5, 5 and 7.5% w w-1) along with stevia were used in chocolate formulation. Nine formulations were examined to determine some physicochemical, mechanical and sensory properties in order to find the optimum concentrations of these components. The lyophilized Lactobacillus paracasei were incorporated in the optimal formulation of prebiotic milk chocolate. The viability of probiotic bacteria in milk chocolate was carried out during storage at 22°C for up to 6 months.
Results and Conclusion: In general, chocolate formulations with high levels of galactooligosaccharide, achieved the highest plastic viscosity and yield stress. The lowest viscosity and yield stress were observed for the samples containing high concentrations of tagatose and in control. In addition, galactooligosaccharide at higher ratios induced the least desirable sensorial effects, whereas tagatose improved the overall acceptability. It can be concluded that the overall acceptability of milk chocolate samples were with (7.5), tagatose: galactooligosaccharide ratios of 2.5%-2.5%, presenting the optimal applicable range as prebiotic compounds. Numbers of live Lactobacillus paracasei cells remained above 8.0 log CFU g-1 until 6 months under ambient conditions. Milk chocolate was shown to be an excellent vehicle for the delivery of Lactobacillus paracasei, and the prebiotic ingredients galactooligosaccharide and tagatose did not interfere in its viability.
Conflict of interest: The authors declare no conflict of interest.
- ▪ Chocolate ▪ Galactooligosaccharide ▪ Prebiotic ▪ Probiotic ▪ Tagatose
How to Cite
References
Hasler CM. Functional foods: their role in disease prevention and health promotion. Food Techn. 1998; 52(11): 63-70.
Milner JA. Functional Foods and Health Promotion. J. Nutr. 1999; 129: (Suppl. 7) 1395s-1397s.
Gallego CG, Salminen S. Novel Probiotics and Prebiotics: How Can They Help in Human Gut Microbiota Dysbiosis? AFB. 2016; 3(2): 72-81.
Rad A. Letter to the editor. Food Chem. 2009; 114: 1073. doi:10.1016/j.foodchem.2008.10.012.
Rad A, Akbarzadeh F, Mehrabany EV. Which are more important: Prebiotics or probiotics? Nutr. 2012; 28: 1196-1197. doi:10.1016/j.nut.2012.03.017.
Laparra JM, Tako E, Glahn RP, Miller DD. Supplemental inulin does not enhance iron bioavailability to Caco-2 cells from milk- or soy-based, probiotic-containing, yogurts but incubation at 37°C does. Food Chem. 2008; 109: 122-128. doi.org/10.1016/j.foodchem.2007.12.027.
Li D, Kim JM, Jin Z, Zhou J. Prebiotic effectiveness of inulin extracted from edible burdock. Anaerobe. 2008; 14: 29-34. doi: 10.1016/j.anaerobe.2007.10.002.
Gibson GR, Williams A, Reading S, Collins MD. Fermentation of non-digestible oligosaccharides by human colonic bacteria. Proc. Nutr. Soc. 1996; 55: 899-912. doi.org/10.1079/PNS19960087.
Homayouni A, Azizi A, Javadi M, Mahdipour S, Ejtahed H. Factors influencing probiotic survival in ice cream: a review. Int. J. Dairy Sci. 2012; 7(1): 1-10. doi: 10.3923/ijds.2012.1.10.
Homayouni Rad AH, Mehrabany EV, Alipoor B, Mehrabany LV, Javadi M. Do probiotics act more efficiently in foods than in supplements? Nutr. 2012; 28(7): 733-736. doi:10.1016/j.nut.2012.01.012.
Rad A, Torab R, Mortazavian AM, Mehrabany EV, Mehrabany, LV. Can probiotics prevent or improve common cold and influenza? Nutr. 2013; 29(5): 805-806. doi: http://dx.doi.org/10.1016/j.nut.2012.10.009.
Rad A, Torab R, Ghalibaf M, Norouzi S, Mehrabany EV. Might patients with immune-related diseases benefit from probiotics? Nutr. 2013; 29(3): 583-586. DOI:10.1016/j.nut.2012.10.008.
Homayouni A, Payahoo L, Azizi A. Effects of probiotics on lipid profile: A review. Am. J. Food Techn. 2012; 7(5): 251-265. doi: 10.3923/ajft.2012.251.265.
Homayoni Rad A, Vaghef Mehrabany E, Alipoor B, Vaghef Mehrabany L. The comparison of food and supplement as probiotic delivery vehicles. Crit. Rev. food Sci. Nutr. 2016; 56(6): 896-909. doi: 10.1080/10408398.2012.733894.
Ejtahed HS, Mohtadi Nia J, Homayouni Rad A, Niafar M, Asghari Jafarabadi M, Mofid V. The effects of probiotic and conventional yoghurt on diabetes markers and insulin resistance in type 2 diabetic patients: a randomized controlled clinical trial. IJEM. 2011; 13(1): 1-8.
Mitsuoak T, Hidaka H, Eida T. Effect of fructo-oligosaccharides on intestinal microflora. Nahrung. 1987; 31: 427-436.
Yazawa K, Imai K, Tamura Z. Oligosaccharides and polysaccharides specifically utilizable by bifidobacteria. Chem. Pharm. Bull. 1978; 26(11): 3306-3311. doi.org/10.1248/cpb.26.3306.
Tomomatsu H. Health effects of oligosaccharides. Food Tech. 1994; 48: 61-64.
Depeint F, Tzortzis G, Vulevic J, I'Anson K, Gibson GR. Prebiotic evaluation of a novel galactooligosaccharide mixture produced by the enzymatic activity of Bifidobacterium bifidum NCIMB 41171, in healthy humans: a randomized, double-blind, crossover, placebo-controlled intervention study. Am. J. Clin. Nutr. 2008; 87(3): 785-91.
Oh DK. Tagatose: properties, applications and biotechnological processes. Appl. Microbiol. Biotechnol. 2007; 76(1): 1-8. doi: 10.1007/s00253-007-0981-1.
Chetana R, Reddy SRY, Negi PS. Preparation and properties of probiotic chocolate using yoghurt powder. Food Nutr. Sci. 2013; 4: 276-281. http://dx.doi.org/10.4236/fns.2013.43037 P.
Nebesny E, Zyzelewicz D, Motyl I, Libudzisz Z. Properties of sucrose-free chocolates enriched with viable lactic acid bacteria. Eur. Food Res. Tech. 2005; 220: 358-362
Patel P, Parekh T, Subhash R. Development of probiotic and symbiotic chocolate mousse: A functional food. Biotech. (Reading, Mass.). 2008; 7(4): 769-774. DOI: 10.3923/biotech.2008.769.774.
Kemsawasd V, Chaikham P, Rattanasena P. Survival of immobilized probiotics in chocolate during storage and with an in vitro gastrointestinal model. Food Biosci. 2016; 16; 37-43. https://doi.org/10.1016/j.fbio.2016.09.001.
Beards E, Tuohy K, Gibson G. A human volunteer study to assess the impact of confectionery sweeteners on the gut microbiota composition. Br. J. Nutr. 2010; 104: 701-708. doi: 10.1017/S0007114510001078.
Suter A. The effect of galactooligosaccharide addition to a chocolate system. A Thesis. The Ohio State University. 2010.
Shourideh M, Taslimi A, Azizi MH, Mohammadifar MA. Effects of D-Tagatose and inulin on some physicochemical, rheological and sensory properties of dark chocolate. Int. J. Biosci. Biochem. Bioinforma. 2012; 2(5): 314-319. doi: 10.7763/IJBBB.2012.V2.124.
Hajipour N, Homayouni Rad A. Response to the paper ‘‘The effect of prebiotics on the viability of encapsulated probiotic bacteria’’. LWT. 2018; 90: 606. DOI: 10.1016/j.lwt.2017.11.035.
Rasouli Pirouzian H, Peighambardoust SH, Azadmard-Damirchi S. Sucrose-free milk chocolate sweetened with different bulking agents: effects on physicochemical and sensory properties. BFAIJ. 2016; 8(2): 340-349.
Nebesny E, Zyzelewicz D, Motyl I, Libudzisz Z. Dark chocolates supplemented with Lactobacillys strains. Eur. Food Res. Tech. 2007; 225: 33-42.
Konar N. Influence of conching temperature and some bulk sweeteners on physical and rheological properties of prebiotic milk chocolate containing inulin. Eur. Food Res. Techn. 2013; 236(1): 135-143. doi: 10.1007/s00217-012-1873-x.
ICA. Determination of moisture content of Cocoa and Chocolate products, Analytical method 26. CAOBISCO, rue Defacqz 1, B-1000 Bruxelles, Belgium. 1988.
Afoakwa E, Paterson A, Fowler M, Vieira J. Comparison of rheological models for determining dark chocolate viscosity. Int. J. Food Sci. Tech. 2009; 44(1): 162-167. doi: 10.1111/j.1365-2621.2008.01710.x.
Abbasi S, Farzanmehr H. Optimization of the formulation of prebiotic milk chocolate based on rheological properties. Food Techn. Biotechnol. 2009; 47(4): 396-403.
Yeganehzad S, Mazaheri Tehrani M, Mohebbi M, Habibi Najafi MB. Effects of replacing skim milk powder with soy flour and ball mill refining time on particle size and rheological properties of compound chocolate. JAST. 2013; 15(1): 125-135.
Afoakwa E, Paterson A, Fowler M, Vieira J. Particle size distribution and compositional effects on textural properties and appearance of dark chocolates. J. Food Eng. 2008; 87(2): 181–190. doi.org/10.1016/j.jfoodeng.2007.11.025.
Popov-Raljic JV, Lalicic-Petronijivic JG. Sensory properties and colour measurements of dietary chocolates with different compositions during storage for up to 360 days. Senors. 2009; 9(3): 1996-2016. doi: 10.3390/s90301996.
Belscak-Cvitanovix A, Komes D, Dujomovic M, Karlovic S, Biskic M, Brncic M, Jezek D. Physical, bioactive and sensory quality parameters of reduced sugar chocolates formulated with natural sweeteners as sucrose alternatives. Food Chem. 2015; 167:61-70. doi.org/10.1016/j.foodchem.2014.06.064.
Lalicˇic´-Petronijevic´ J, Popov-Raljic´ J, Obradovic´ D, Radulovic´ Z, Paunovic´ D, Petrušic´ M, Pezo L. Viability of probiotic strains Lactobacillus acidophilus NCFM_ and Bifidobacterium lactis HN019 and their impact on sensory and rheological properties of milk and dark chocolates during storage for 180 days. J. Funct. Foods. 2015; 15: 541-550. https://doi.org/10.1016/j.jff.2015.03.046.
Anonymous, Gaio®tagatose in chocolate. [Online]. Available: http://www.gaio- tagatose.com. 2004.
Torres Duarte PM, Gonçalves Maria do Pilar F, Teixeira José A, Rodrigues Lígia R. Galacto-Oligosaccharides: Production, Properties, Applications, and Significance as Prebiotics. CRFSFS. 2010; 9(5): 438-454. doi: 10.1111/j.1541-4337.2010.00119.x.
IOCCC. Viscosity of chocolate. Determination of Casson yield value and Casson plastic viscosity. 1973.
Bouzas J, Brown BD. Ingredient Interactions. Marcel and Dekker, New York, 1995: 451-528.
Aeschlimann JM, Beckett ST. International inter-laboratory trials to determine the factors affecting the measurement of chocolate viscosity. J. Texture Stud. 2000; 31(5): 541-576. DOI: 10.1111/j.1745-4603.2000.tb01019.x.
Rasouli Pirouzian H, Peighambardoust SH, Azadmard-Damirchi S. Rheological Properties of Sugar-Free Milk Chocolate: Comparative Study and Optimisation. Czech J. Food Sci. 2017; 35(5): 440-448. doi: 10.17221/231/2016-CJFS.
Saputro AD, Walle DV, Aidoo RP, Mensah MA, Delbaere C, Clercq ND, Durme JV, Dewettinck K. Quality attributes of dark chocolates formulated with palm sap-based sugar as nutritious and natural alternative sweetener. Eur. Food Res. Tech. 2017; 243(2): 177-191. doi:10.1007/s00217-016-2734-9.
Beckett ST. Industrial Chocolate Manufacture and Use (3rd ed). Blackwell Publishing, Oxford, 1999: 686.
Jackson K. Recipes. Blackwell Science, Oxford, London, 1999: 323-346. Industrial chocolate manufacture and use (Ed. S.T. Beckett, 3rd ed).
Ziegler GR, Mongia G, Hollender R. Role of particle size distribution of suspended solids in defining the sensory properties of milk chocolate. Int. J. Food Prop. 2001; 4(2): 353-370. doi.org/10.1081/JFP-100105199.
Gibson GR, Beatty E, Wang X, Cummings JH. Selective stimulation of bifidobacteria in the human colon by oligofructose and inulin. Gastroenteology. 1995; 108; 975-982.
Crittenden R. Incorporating probiotics into foods. In Y. K. Lee & S. Salminen (Eds.), Handbook of probiotics and prebiotics (pp. 58-75). Hoboken, 2009, NJ: JohnWiley & Sons, Inc.
Homayouni Rad A, Javadi M, Ghasemnezhad Tabrizian V, Alizadeh M. A survey to increasing the probiotic survival in functional ice cream by microencapsulation. J. Food Res. (Agricultural Sci.). 2012; 21(3): 97-102.
Homayouni A, Ehsani MR, Azizi A, Razavi SH, Yarmand MS. Growth and survival of some probiotic strains in simulated ice cream conditions. J. Appl Sci. 2008; 8(2): 379-382. DOI: 10.3923/jas.2008.379.382.
Silva MP, Tulini FL, Marinho JFU, Mazzocato MC, De Martinis ECP, Luccas V, Favaro-Trindade CS. Semisweet chocolate as a vehicle for the probiotics Lactobacillus acidophilus LA3 and Bifidobacterium animalis subsp. lactis BLC1: Evaluation of chocolate stability and probiotic survival under in vitro simulated gastrointestinal conditions. LWT. 2017; 75: 640-647. https://doi.org/10.1016/j.lwt.2016.10.025.
Zyzelewicz D, Nebesny E, Motyl I, Libudzisz Z. Effect of milk chocolate supplementation with lyophilized Lactobacillus cells on its attributes. CJFS. 2010; 28(5): 392-406.
Aragon-Alegro LC, Alarcon-Alegro JH, Cardarelli HR., Chiu MC, Saad SMI. Potentially probiotic and synbiotic chocolate mousse. LWT. 2007; 40: 669-675. https://doi.org/10.1016/j.lwt.2006.02.020.
- Abstract Viewed: 1354 times
- PDF Downloaded: 609 times